Aug . 29, 2025 00:40 Back to list
Nylon Flocked Nasal Swab for Comfortable Testing
Industry Trends and the Evolving Role of Nasal Swabs
The global medical diagnostics landscape has undergone a profound transformation, driven by advancements in biotechnology, increasing prevalence of infectious diseases, and a heightened focus on early and accurate detection. At the forefront of this evolution is the ubiquitous nasal swab, an indispensable tool for specimen collection. Its critical role has been underscored by recent public health imperatives, accelerating innovation in materials science, manufacturing precision, and collection efficacy.
Current industry trends highlight a move towards enhanced patient comfort, superior sample yield, and standardized, reliable collection methods. The demand for high-quality nasal test swab solutions, particularly those utilizing advanced materials like nylon flocked swabs, is expanding rapidly across clinical diagnostics, epidemiological surveillance, and research & development sectors. This shift is driven by the need for diagnostic tools that can consistently deliver optimal viral and bacterial load collection, crucial for the sensitivity of downstream molecular assays such as PCR and antigen testing. Moreover, the integration of automation in sample processing further emphasizes the need for swabs with consistent break-points and minimal inhibitory substances.
Key market drivers include the rising demand for point-of-care testing, the continuous threat of new pathogens, and the global expansion of healthcare infrastructure. Manufacturers are responding by focusing on sterile, individually packaged products that meet rigorous international quality standards, ensuring both safety and performance across diverse application environments.
Technical Specifications and Product Overview: Disposable Sterile Nasal Swab
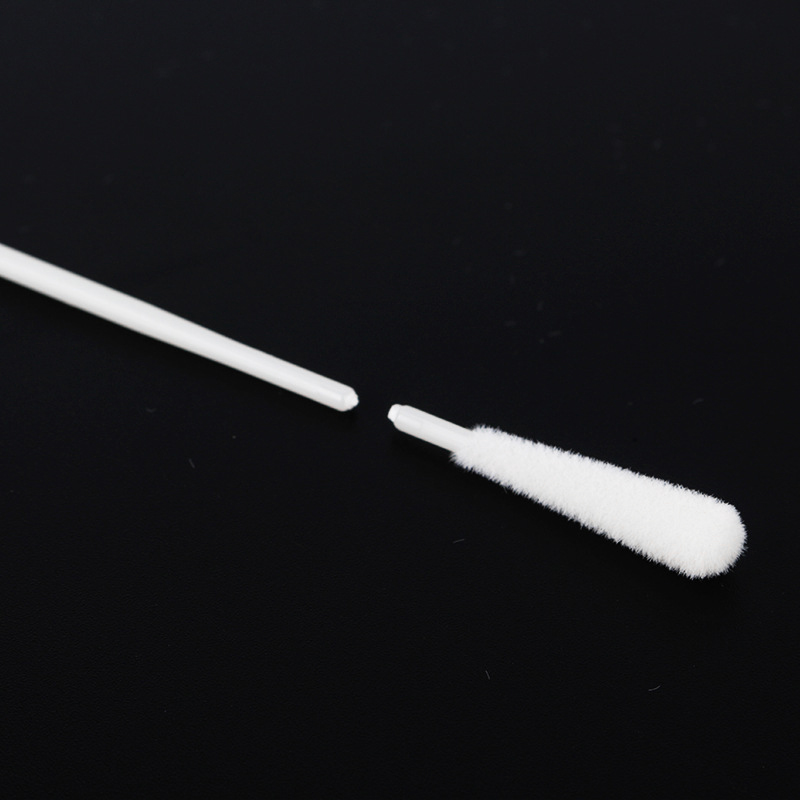
The Disposable Sterile Nasal Swab is engineered for optimal collection of clinical specimens from the nasopharynx and oropharynx. Designed with precision, these swabs prioritize both collection efficiency and patient comfort, crucial for high-volume testing scenarios.
At the core of its design is the nylon flocked swabs tip, which features a perpendicular nylon fiber structure. This unique design acts like a soft brush, effectively collecting a high volume of cellular material and liquid samples. The capillary action between the fibers facilitates strong hydraulic absorption, allowing for superior sample elution into transport media compared to traditional spun fiber swabs. The handle is typically made from medical-grade ABS plastic, designed with an engineered breakpoint to allow for safe and easy insertion into specimen tubes.
Key Technical Parameters:
| Parameter | Specification |
|---|---|
| Swab Tip Material | Medical-grade Nylon Flocked Fibers |
| Handle Material | Medical-grade ABS Plastic |
| Overall Length | Typically 150mm (Nasal) / 142mm (Oropharyngeal) |
| Break-point Design | Pre-scored at 80mm from tip (standard) |
| Sterilization Method | Ethylene Oxide (EO) or Gamma Irradiation |
| Packaging | Individual peel-pouch, sterile |
| Certifications | ISO 13485, CE Marked, FDA Registered |
| Shelf Life | Typically 3 years from manufacturing date |
These specifications ensure that each nasal swab performs consistently, minimizing pre-analytical errors and maximizing the reliability of diagnostic results. The flocked tip's design allows for rapid elution of the entire sample, including epithelial cells and viral particles, into transport media, ensuring a high quality specimen for subsequent molecular analysis.

Manufacturing Process Flow for Nasal Swabs
The production of a high-quality Disposable Sterile Nasal Swab is a meticulously controlled, multi-stage process, adhering to stringent quality management systems like ISO 13485. This ensures each swab meets the demanding performance and safety standards required for medical devices.
Detailed Process Steps:
1. Material Procurement & Inspection:
Raw materials, including medical-grade nylon fibers, ABS plastic granules, and peel-pouch materials, are sourced from validated suppliers. Each batch undergoes rigorous incoming quality control (IQC) to verify compliance with purity, biocompatibility, and physical property specifications. (e.g., ISO 10993 for biocompatibility).
2. Handle Molding (Casting/Injection Molding):
ABS plastic granules are injection molded into precise handle forms, incorporating the engineered break-point. This process (akin to casting/forging in other industries) requires high-precision tooling and controlled temperature/pressure parameters to ensure consistent dimensions and mechanical strength, crucial for the swab's structural integrity and safe use.
3. Flocking Process:
Nylon fibers are electrostatically applied onto the molded handle tip. This specialized technique ensures perpendicular orientation and dense packing of the fibers, creating the superior collection surface characteristic of nylon flocked swabs. Precise control of flocking parameters is essential for optimal sample collection and release.
4. Drying & Curing:
After flocking, the swabs undergo a controlled drying and curing process to firmly bond the fibers to the handle, preventing shedding and ensuring durability during sample collection.
5. Assembly & Packaging (Cleanroom Environment):
Swabs are inspected for visual defects and then individually sealed into sterile peel-pouches in an ISO Class 8 or higher cleanroom environment. This step prevents particulate contamination and maintains product integrity prior to sterilization.
6. Sterilization:
Packaged swabs undergo terminal sterilization, typically using Ethylene Oxide (EO) gas or Gamma Irradiation. Sterilization cycles are validated according to ISO 11135 (for EO) or ISO 11137 (for Gamma) to achieve a Sterility Assurance Level (SAL) of 10-6, meaning less than one in a million chance of a non-sterile product.
7. Final Quality Control & Release:
Post-sterilization, a final quality check confirms package integrity, sterility (through biological indicator testing), and absence of residual sterilants. Products are released only after all testing standards (e.g., ISO 13485, ANSI/AAMI ST79) are met. The expected service life of these swabs, typically stored at ambient conditions, is 3 years.
This meticulous process ensures that each disposable sterile nasal swab is of the highest quality, ready for critical diagnostic applications in target industries like clinical diagnostics, public health, and research.
Technical Advantages and Application Scenarios
The design and manufacturing superiority of modern nasal test swab products provide distinct technical advantages that translate directly into improved diagnostic outcomes and operational efficiencies.
Key Technical Advantages:
- Superior Sample Collection: The flocked tip's unique structure with millions of perpendicular microfibers maximizes surface area for efficient collection of cells and viral particles, outperforming traditional spun-fiber swabs by up to 3x in sample yield, crucial for detecting low viral loads.
- Enhanced Sample Elution: Unlike traditional swabs where samples can get trapped in the fiber matrix, the flocked design allows for near-complete (typically >95%) elution of the collected specimen into the transport medium, ensuring more target analyte is available for testing.
- Patient Comfort & Safety: The soft, flexible nylon tip minimizes discomfort during collection, leading to better patient compliance. The medical-grade materials are non-toxic and non-inhibitory to PCR, safeguarding diagnostic assay performance.
- Integrated Break-point: The pre-scored handle allows for easy and aseptic breaking of the swab into a transport tube, reducing contamination risk and improving workflow efficiency in high-throughput laboratories.
- Sterility Assurance: Each swab is individually packaged and terminally sterilized, guaranteeing sterility at the point of use, which is paramount for preventing cross-contamination in diagnostic procedures.
Application Scenarios:
These disposable sterile swabs find extensive use across various critical application scenarios:
- Clinical Diagnostics: Essential for collecting respiratory specimens for the detection of viral pathogens (e.g., influenza, RSV, SARS-CoV-2) and bacterial pathogens (e.g., Streptococcus pneumoniae, Bordetella pertussis) using PCR, RT-PCR, and antigen rapid tests.
- Epidemiological Surveillance: Utilized in public health initiatives for widespread testing and monitoring of infectious disease outbreaks, providing crucial data for pandemic preparedness and response.
- Research & Development: Used in virology, bacteriology, and immunology research for collecting clinical samples for novel assay development, vaccine efficacy studies, and pathogen characterization.
- Veterinary Diagnostics: Applied in animal health for the diagnosis of respiratory diseases in livestock and companion animals, reflecting cross-species diagnostic needs.
- Environmental Monitoring: In specialized applications, sterile swabs can be used for surface sampling in controlled environments to monitor microbial contamination, demonstrating versatility beyond direct patient care.
These diverse applications underscore the versatility and critical importance of high-performance nasal swab technology in modern healthcare and scientific endeavors.

Vendor Comparison and Customized Solutions
Selecting the right vendor for nasal test swab products is a strategic decision for healthcare providers, diagnostic laboratories, and research institutions. While many suppliers offer sterile swabs, differentiation lies in product quality, manufacturing compliance, customization capabilities, and post-sales support.
Key Factors for Vendor Comparison:
| Feature/Criterion | Standard Vendor Offerings | Prisesbio (Example of Premium Vendor) |
|---|---|---|
| Flocked Tip Technology | Often basic nylon flocking; variable density. | High-density, perpendicular nylon flocking; optimized for maximum sample collection & release. |
| Handle Material & Break-point | Standard PP/PS; inconsistent break-points. | Medical-grade ABS; precise, clean break-point for automation compatibility. |
| Sterilization & Certifications | EO sterilization; basic local certifications. | Validated EO or Gamma sterilization; ISO 13485, CE, FDA Registered, MDSAP. |
| DNase/RNase Free & PCR Inhibitor Free | Not always guaranteed or tested. | Routinely tested and certified free, ensuring molecular assay compatibility. |
| Customization Options | Limited to standard sizes/packaging. | Extensive customization for length, break-point, tip size, packaging, and branding. |
| Technical Support & Warranty | Basic support, standard warranty. | Dedicated technical team, comprehensive warranty, responsive after-sales. |
Customized Solutions: Tailoring the Nasal Swab to Specific Needs
Recognizing that diverse diagnostic workflows and research protocols often require specialized tools, leading manufacturers offer extensive customization capabilities for their nasal swab product lines. This approach allows clients to optimize their sample collection for specific applications, enhancing efficiency and accuracy.
- Swab Dimensions: Custom lengths for both the flocked tip and the handle can be engineered to suit specific anatomical considerations (e.g., pediatric vs. adult) or specialized collection sites.
- Break-point Configuration: While a standard 80mm break-point is common, custom break-points can be designed for compatibility with automated sample processing systems or specific transport tube dimensions.
- Tip Size & Shape: Variations in tip diameter and shape can be developed to optimize collection for different viscosities of bodily fluids or specific epithelial cell types.
- Packaging & Branding: Customized primary (peel-pouch) and secondary packaging (boxes) with client-specific branding, labeling, and lot tracking can be implemented for seamless integration into existing supply chains.
- Sterilization Method: While EO is standard, gamma irradiation or other sterilization methods can be employed based on client preference or specific material compatibility requirements.
These tailored solutions exemplify a commitment to partnership, ensuring that the diagnostic tools precisely align with the evolving needs of advanced medical and research environments.

Application Case Studies and Customer Experience
The real-world impact of high-quality nylon flocked swabs is best demonstrated through their successful deployment in demanding clinical and research environments. These case studies highlight the tangible benefits experienced by end-users.
Case Study 1: Large-Scale Public Health Screening Program
A national public health agency implemented a mass screening program for respiratory pathogens across its populace. Facing the challenge of collecting millions of specimens reliably, they partnered with a vendor specializing in high-performance nasal swab products. Over 10 million disposable sterile nasal swabs were supplied within a six-month period. Post-implementation data showed a significant reduction in "insufficient sample" rejections from diagnostic labs, dropping from an average of 3.5% to below 0.8%. This improvement was directly attributed to the superior sample collection and elution properties of the flocked swabs, leading to enhanced diagnostic accuracy and faster turnaround times for critical health decisions.
Case Study 2: Hospital System Adopting Automation
A large hospital network sought to automate its pre-analytical sample processing to manage increasing test volumes. A critical component was the compatibility of collection devices with their robotic un-capping and sample aliquoting systems. After evaluating several manufacturers, they selected a customized nasal test swab with a precisely engineered break-point and consistent handle dimensions. This choice enabled seamless integration into their automated workflow, drastically reducing manual handling errors and associated labor costs. The hospital reported a 25% increase in daily sample processing capacity and a 15% reduction in total turnaround time for respiratory panel testing, demonstrating the efficiency advantages in typical application scenarios.
Customer Feedback Highlights:
- "The consistency of the break-point is outstanding. Our automated systems never miss, which saves us hours every day." - Lab Manager, Major Diagnostic Reference Laboratory
- "Patients consistently report less discomfort with these swabs compared to previous brands, which is a huge plus for compliance, especially in pediatric cases." - Infectious Disease Clinician, University Medical Center
- "We've seen a noticeable improvement in our PCR cycle threshold values, indicating higher viral load recovery. This helps us catch even early-stage infections." - Virology Researcher, Biomedical Institute

Ensuring Trustworthiness and Authoritativeness (Google )
Building trust and establishing authority are paramount in the B2B medical device sector. Our commitment to excellence is demonstrated through rigorous adherence to international standards, transparent processes, and unwavering customer support. For over a decade, we have been a trusted partner to healthcare providers globally, supplying millions of high-quality medical devices.
Certifications and Authoritative References:
- ISO 13485:2016 Certified: Our manufacturing facilities and quality management systems are certified to ISO 13485, the international standard for medical device quality systems, ensuring consistent product quality and regulatory compliance.
- FDA Registered: Our products are registered with the U.S. Food and Drug Administration, affirming compliance with US regulatory requirements for medical devices.
- CE Marked: Products conform to European health, safety, and environmental protection standards, enabling distribution within the European Economic Area.
- MDSAP Certified: Medical Device Single Audit Program certification demonstrates compliance with regulatory requirements of multiple countries (e.g., USA, Canada, Brazil, Australia, Japan).
- Biocompatibility Testing (ISO 10993): All materials used in our nasal swab products undergo extensive biocompatibility testing to ensure safety for human contact.
We partner with leading diagnostic companies and healthcare systems worldwide, a testament to our product reliability and service excellence. Our dedication to quality is reflected in comprehensive internal test data, showing consistent performance metrics for sample collection efficiency and elution rates.

Frequently Asked Questions (FAQ)
Q: Are your nasal swabs compatible with all viral transport media (VTMs)?
A: Our disposable sterile nasal swabs are designed for broad compatibility with most commercially available VTMs and saline solutions. The nylon flocked tip ensures optimal elution into aqueous solutions without inhibiting downstream molecular assays. We recommend verifying compatibility with specific VTMs if unique formulations are used.
Q: What is the typical lead time for a bulk order?
A: Standard bulk orders (e.g., 500,000 units) typically have a lead time of 2-4 weeks, depending on current production schedules and customization requirements. For larger or highly customized orders, lead times will be provided upon quotation. We maintain significant inventory to support urgent demands and streamline logistics for our B2B partners.
Q: What is your warranty policy for disposable sterile nasal swabs?
A: We offer a comprehensive warranty covering manufacturing defects for the entire stated shelf life of the product (typically 3 years from the date of manufacture). This warranty ensures that our products meet all specified performance and quality standards under normal storage and usage conditions.
Q: How can I obtain technical support or request a custom solution?
A: Our dedicated customer support and technical teams are available to assist with any inquiries. You can reach us via phone, email, or through our website's contact form. For custom solutions, our R&D specialists will work closely with you from concept to production to ensure your exact specifications are met. Visit our 'Contact Us' page for detailed information.
Conclusion
The strategic importance of high-quality disposable sterile nasal swabs in modern diagnostics cannot be overstated. As the first critical step in many diagnostic workflows, the performance of the nasal swab directly influences the accuracy and reliability of downstream testing. By focusing on advanced materials, precision manufacturing, rigorous quality control, and comprehensive customer support, we continue to deliver solutions that meet the evolving demands of the global healthcare and research communities, ensuring optimal sample collection and reliable diagnostic outcomes.
References
1. World Health Organization. (2020). Laboratory testing for coronavirus disease 2019 (COVID-19) in suspected human cases. Interim guidance.
2. Centers for Disease Control and Prevention. (2021). Interim Guidelines for Collecting, Handling, and Testing Clinical Specimens for COVID-19.
3. Clinical and Laboratory Standards Institute. (2018). Collection, Transport, Preparation, and Storage of Specimens for Molecular Methods; Approved Guideline—Third Edition (MM3-A3).
4. ISO 13485:2016. (2016). Medical devices — Quality management systems — Requirements for regulatory purposes.
5. European Medicines Agency. (2021). Guidance on the use of CE-marked medical devices in clinical trials.
-
Buy at home covid test rapid covid test kit antigen test covid19
NewsApr.16,2026
-
Covid19 Antigent Test Kit Cassette Covid Test Rapid Diagnostic Covid 19 Antigen Rapid Test
NewsApr.09,2026
-
Comprehensive Guide to Antigen Covid Test Covid Saliva Test and Covid Test Kit 19 Options
NewsApr.07,2026
-
Comprehensive Guide to Test Kit Covid Covid19 Rapid Antigen Test Self Test Covid Options
NewsApr.04,2026
-
Comprehensive Guide to Rapid Test Kits COVID19 for Accurate Covid Self Test Results
NewsMar.31,2026
-
Comprehensive Guide to Rapid Antigen Test Kit Covid 19 Covid Rapid Test Kit Test Kit Covid 19
NewsMar.28,2026

