Sep . 09, 2025 19:40 Back to list
Accurate HIV Ab/Ag Gen Blood Rapid Test Kit - 4th Gen Detection
Advanced HIV Diagnostics: The Role of 4th Generation Rapid Tests
In the global effort to combat HIV/AIDS, timely and accurate diagnosis is paramount for effective treatment initiation and prevention strategies. Fourth-generation HIV rapid diagnostic tests represent a significant advancement, offering a shorter diagnostic window by simultaneously detecting both HIV-1 p24 antigen and antibodies to HIV-1 and HIV-2. Among these, the hiv ab/ag gen blood rapid test kit stands out as a crucial tool for accelerating diagnosis, particularly in point-of-care settings where immediate results can drastically improve patient outcomes and public health management.
This comprehensive overview delves into the critical aspects of advanced HIV diagnostics, focusing on the technological prowess and operational advantages of the 4th generation rapid test kits designed for blood samples. We will explore the latest industry trends, delve into the intricate technical specifications, highlight diverse application scenarios, and underscore the inherent advantages that position these kits as indispensable assets in modern healthcare.
Industry Trends and the Evolving Landscape of HIV Diagnostics
The landscape of HIV diagnostics is continually evolving, driven by the imperative for earlier detection, enhanced accessibility, and simplified testing protocols. A significant trend is the shift from laboratory-centric testing to decentralized, point-of-care (POC) solutions. This move is largely fueled by the success of products like the hiv ab/ag rapid test, which enables testing outside traditional clinical laboratory settings, bringing diagnostics closer to the patient. This is particularly vital in resource-limited environments and for reaching key populations.
Fourth-generation rapid tests have emerged as the gold standard for initial screening due to their ability to detect HIV infection significantly earlier than antibody-only tests. By simultaneously targeting the HIV-1 p24 antigen and anti-HIV antibodies (IgM and IgG), these kits reduce the diagnostic window period from several weeks (for antibody-only tests) to as little as 2-4 weeks post-exposure. This early detection capability is critical for preventing onward transmission and facilitating prompt linkage to care. Furthermore, there's a growing emphasis on integrated diagnostic platforms, digital health connectivity for data reporting, and increased automation to minimize human error and scale up testing capacities globally.
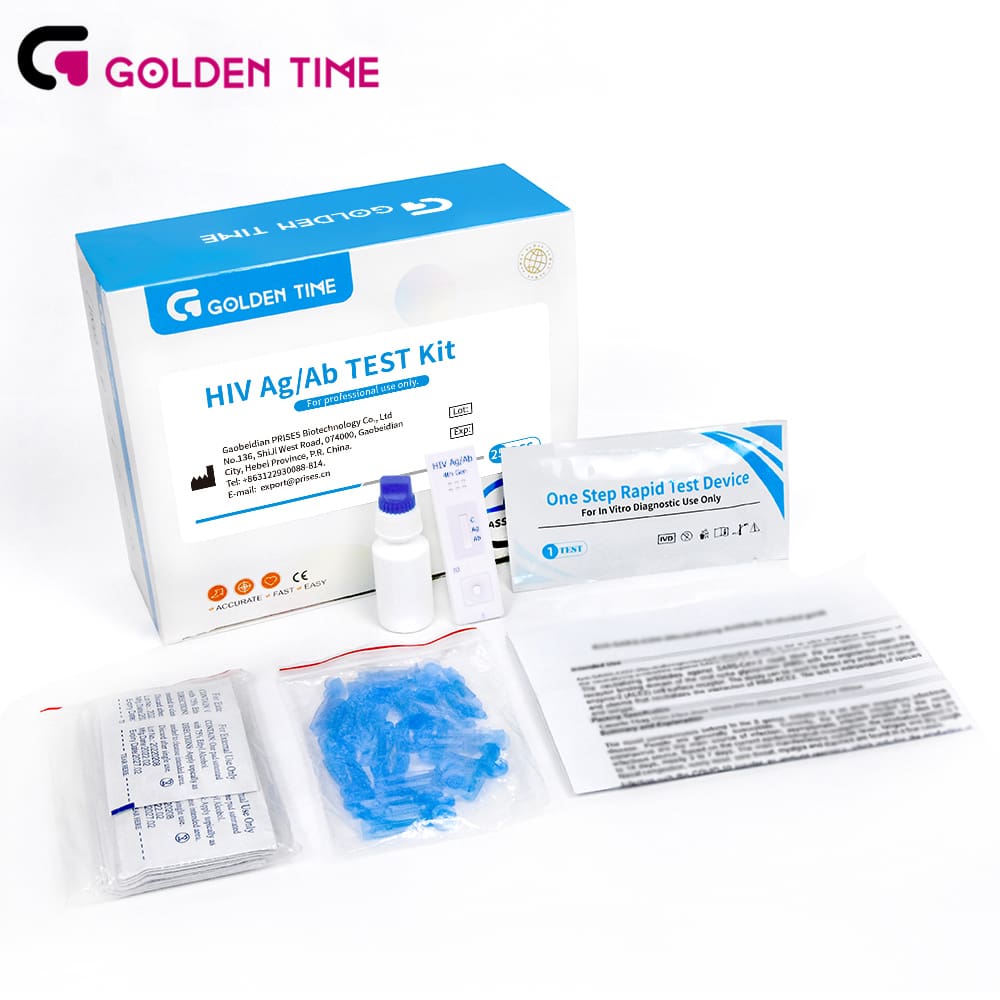
Technical Specifications and Operating Principles of the HIV Ab/Ag 4th Gen Blood Rapid Test Kit
The hiv ab/ag gen blood rapid test kit is a highly sophisticated in vitro diagnostic device utilizing immunochromatographic technology. It is designed to qualitatively detect the HIV-1 p24 antigen, as well as antibodies to HIV-1 (subtypes O/M/N) and HIV-2 in human whole blood, serum, or plasma specimens. This dual-target approach significantly enhances its diagnostic utility.
Operating Principle:
The test operates on the principle of lateral flow immunoassay. The test cassette contains a sample pad, a conjugate pad, a nitrocellulose membrane with specific test (T) lines and a control (C) line, and an absorbent pad. The conjugate pad is impregnated with colloidal gold conjugated anti-p24 antibodies and recombinant HIV antigens (HIV-1, HIV-2, and HIV-1 O/M/N synthetic peptides). The T-line for antigen detection contains immobilized anti-p24 antibodies, while the T-line for antibody detection contains immobilized recombinant HIV antigens. The C-line contains goat anti-mouse IgG antibodies to capture excess colloidal gold conjugates, ensuring proper test function.
When a treated specimen is applied to the sample pad, it migrates by capillary action. If HIV-1 p24 antigen is present, it binds to the gold-conjugated anti-p24 antibodies, forming an immunocomplex that then binds to the immobilized anti-p24 antibodies at the antigen T-line, producing a visible colored line. Simultaneously, if HIV-1 or HIV-2 antibodies are present, they bind to the gold-conjugated HIV antigens, forming another immunocomplex that binds to the immobilized HIV antigens at the antibody T-line, producing a second colored line. The control line should always appear, confirming the validity of the test.
Key Specifications (PrisesBio HIV Ab/Ag 4th Gen Blood Rapid Test Kit):
| Parameter | Specification |
|---|---|
| Product Name | HIV Ab/Ag 4th Gen Blood Rapid Test Kit |
| Detection Target | HIV-1 p24 antigen, HIV-1 antibodies (IgM/IgG), HIV-2 antibodies (IgM/IgG) |
| Sample Type | Whole Blood, Serum, Plasma |
| Detection Principle | Immunochromatography (Lateral Flow Assay) |
| Test Time | 15-20 minutes |
| Sensitivity | >99.5% (Based on clinical evaluations) |
| Specificity | >99.0% (Based on clinical evaluations) |
| Storage Temperature | 2-30°C (36-86°F) |
| Shelf Life | 24 months from manufacturing date |
| Regulatory Compliance | CE, ISO 13485 |
Manufacturing Process Flow of the HIV Ab/Ag 4th Gen Blood Rapid Test Kit
The production of a high-quality hiv ab/ag gen blood rapid test kit involves a stringent, multi-step manufacturing process, adhering to international quality management systems like ISO 13485. This ensures the consistent performance, reliability, and safety of each kit. Unlike industrial components that involve casting or machining, diagnostic kits require precise biochemical engineering and meticulous assembly.
Process Stages:
-
Reagent Preparation and Conjugation:
- Synthesis and purification of recombinant HIV antigens (for antibody detection) and high-affinity monoclonal anti-p24 antibodies (for antigen detection).
- Conjugation of selected antibodies and antigens to colloidal gold nanoparticles, forming the detectable signal component. This step is critical for sensitivity.
-
Membrane Coating and Stripping:
- High-quality nitrocellulose membranes are precisely dispensed with specific capture reagents: anti-p24 antibodies for the antigen test line, and recombinant HIV antigens for the antibody test line.
- A control line (C-line) is coated with anti-mouse IgG antibodies to ensure proper test procedure and reagent functionality.
- The membranes are then dried under controlled environmental conditions.
-
Pad Preparation (Sample, Conjugate, Absorbent):
- The conjugate pad is treated with the colloidal gold conjugates and dried.
- Sample and absorbent pads are prepared to ensure optimal sample flow and absorption.
-
Cassette Assembly:
- The prepared membrane strip, conjugate pad, sample pad, and absorbent pad are precisely assembled into a plastic test cassette housing. This step is often automated to ensure consistency.
-
Quality Control (QC) & Validation:
- Each batch undergoes rigorous QC testing for sensitivity, specificity, reproducibility, stability, and absence of cross-reactivity with common interfering substances.
- Performance is verified against established positive and negative control panels, often including seroconversion panels.
-
Packaging and Labeling:
- Individual test kits are sealed in foil pouches with desiccants to maintain stability.
- Accurate labeling with lot numbers, expiry dates, and regulatory markings (e.g., CE, FDA) is applied.
The entire process is governed by strict Good Manufacturing Practices (GMP) and certified under ISO 13485, ensuring that every hiv ab/ag test kit meets the highest international standards for medical devices. The service life of these kits, typically 24 months, is validated through accelerated and real-time stability studies, ensuring consistent performance over their lifespan when stored correctly.
Application Scenarios and Target Industries
The versatility and rapid nature of the hiv ab/ag rapid test make it suitable for a wide array of application scenarios across various sectors within the healthcare and public health domains. Its design specifically addresses the need for quick, reliable diagnostics where traditional lab infrastructure may be limited or where immediate results are critical.
Key Application Scenarios:
- Point-of-Care (POC) Testing: Ideal for clinics, community health centers, mobile testing units, and emergency rooms where rapid turnaround times are essential for patient management decisions. It allows for immediate counseling and linkage to care.
- Blood Banks and Transfusion Services: While advanced nucleic acid tests (NAT) are primary for blood screening, rapid tests can serve as supplementary screening in emergency situations or resource-limited settings before more definitive testing.
- Antenatal Care (ANC): Pregnant women can be screened efficiently during their first ANC visit, enabling prompt intervention to prevent mother-to-child transmission (PMTCT) if positive.
- Public Health Screening Programs: Large-scale community screening drives, especially in high-prevalence areas or among key populations, benefit immensely from the ease of use and rapid results of these kits.
- Harm Reduction Programs: In settings such as needle exchange programs or drug rehabilitation centers, rapid testing facilitates accessible and non-intrusive screening.
- Post-Exposure Prophylaxis (PEP) Assessment: Expedites baseline testing for individuals who have potentially been exposed to HIV, allowing for timely initiation of PEP regimens.
Target Industries:
- Healthcare Providers: Hospitals, clinics, private practices, and primary care facilities.
- Government Health Agencies: Ministries of Health, national public health institutes, disease control centers.
- Non-Governmental Organizations (NGOs): International and local NGOs involved in HIV/AIDS prevention and care.
- Research Institutions: Clinical trials and epidemiological studies requiring rapid screening tools.
- Emergency Medical Services (EMS): For rapid assessment in critical situations.
In these scenarios, the primary advantages demonstrated by the kit are early detection, simplicity of use, rapid results (15-20 minutes), and minimal infrastructure requirements. This accessibility translates into improved public health outcomes by facilitating earlier diagnosis and intervention.
Technical Advantages of 4th Generation HIV Ab/Ag Rapid Test Kits
The evolution from first to fourth-generation HIV tests marks significant progress in diagnostic capabilities. The hiv ab/ag gen blood rapid test kit offers distinct technical advantages that make it superior for initial HIV screening:
- Reduced Window Period: The most significant advantage is the ability to detect HIV infection much earlier. By simultaneously detecting the p24 antigen, which appears in the blood before antibodies, the diagnostic window period is shortened to approximately 2-4 weeks post-exposure, compared to 3-12 weeks for antibody-only tests. This early detection is crucial for preventing further transmission.
- Dual Detection Mechanism: Combining antigen and antibody detection enhances the sensitivity across different stages of infection. P24 antigen levels are high during the acute phase (before seroconversion), while antibodies become dominant in the later stages. This dual approach ensures comprehensive detection.
- High Sensitivity and Specificity: Modern 4th generation kits exhibit exceptionally high sensitivity (>99.5%) and specificity (>99.0%), minimizing false negatives and false positives, especially when used in appropriate testing algorithms. This reliability is critical for accurate diagnoses.
- Ease of Use and Rapid Results: Requiring minimal training and no specialized equipment, these kits can be easily administered by trained personnel in diverse settings. Results are typically available within 15-20 minutes, facilitating immediate patient management decisions.
- Cost-Effectiveness: Compared to laboratory-based ELISA or Western Blot assays, rapid tests are significantly more cost-effective per test, making large-scale screening programs economically viable, particularly in low-resource settings.
- Stable and Portable: Designed for robust performance in various climates, these kits often have broad storage temperature ranges (2-30°C) and long shelf lives, making them suitable for distribution and use in remote or challenging environments.
These technical advantages collectively position the 4th generation rapid test kits as indispensable tools in global HIV prevention and control efforts, directly contributing to the "95-95-95" targets set by UNAIDS.
Vendor Comparison: Evaluating HIV Ab/Ag Rapid Test Kits
Choosing the right hiv ab/ag test kit involves a critical evaluation of various vendor offerings based on key performance indicators, regulatory compliance, and logistical support. While many manufacturers produce 4th generation rapid tests, their performance characteristics and overall value can differ. Below is a comparative overview, emphasizing the strengths of a leading kit like PrisesBio's against general market offerings.
| Feature/Parameter | PrisesBio HIV Ab/Ag 4th Gen Blood Rapid Test Kit | Competitor A (Representative Market Average) | Competitor B (Premium Market Offering) |
|---|---|---|---|
| Detection Targets | HIV-1 p24 Ag, HIV-1 Ab (IgM/IgG), HIV-2 Ab (IgM/IgG) | HIV-1 p24 Ag, HIV-1/2 Ab | HIV-1 p24 Ag, HIV-1 Ab (IgM/IgG), HIV-2 Ab (IgM/IgG) |
| Sample Type | Whole Blood, Serum, Plasma | Whole Blood, Serum, Plasma | Whole Blood, Serum, Plasma |
| Test Time | 15-20 minutes | 15-30 minutes | 15-20 minutes |
| Sensitivity (Clinical) | >99.5% | 98.5% - 99.0% | >99.5% |
| Specificity (Clinical) | >99.0% | 98.0% - 98.8% | >99.2% |
| Storage Temp. | 2-30°C | 4-30°C | 2-30°C |
| Certifications | CE, ISO 13485 | CE, (some regions) | CE, FDA, ISO 13485, WHO Prequalified |
| Typical Cost-Efficiency | High (Excellent performance/cost ratio) | Moderate | Good (Higher upfront cost, but premium performance) |
PrisesBio's offering consistently demonstrates competitive or superior performance in critical parameters such as sensitivity, specificity, and a broad acceptable storage temperature range, ensuring reliability even in challenging logistical environments. While some premium offerings may boast additional certifications like WHO Prequalification, PrisesBio's adherence to CE and ISO 13485 standards ensures robust quality and regulatory compliance for global markets, providing an optimal balance of performance and accessibility.

Customized Solutions and Application Case Studies
Customized Solutions:
Recognizing the diverse needs of global partners, manufacturers often provide customized solutions for the hiv ab/ag gen blood rapid test kit. These can include:
- Bulk Packaging Options: Tailoring kit configurations and packaging sizes to meet the demands of large-scale public health programs or integrated health systems.
- Private Labeling/OEM Services: Allowing partners to brand the kits under their own label, facilitating market penetration and brand building in specific regions.
- Regional Regulatory Adaptation: Assisting with documentation and compliance for specific national regulatory approvals beyond standard CE or ISO, such as FDA EUA for certain markets, or local Ministry of Health registrations.
- Training and Implementation Support: Providing comprehensive training materials and on-site support for healthcare workers to ensure proper test administration and result interpretation, particularly in new deployment settings.
Application Case Studies & Customer Experience:
Case Study 1: National HIV Screening Program in Sub-Saharan AfricaA large-scale national HIV screening program was launched in a Sub-Saharan African country, aiming to increase diagnosis rates among high-risk populations and in rural areas with limited lab access. The program adopted PrisesBio's HIV Ab/Ag 4th Gen Blood Rapid Test Kits for their ease of use, rapid results, and high reliability in field conditions. Over 500,000 tests were conducted in community outreach and mobile clinics within a year. The kits’ ability to detect early infections (p24 antigen) proved invaluable, identifying cases that would have been missed by antibody-only tests, thus allowing earlier intervention and reducing onward transmission. Customer feedback highlighted the kit’s robust performance, clear result interpretation, and minimal training requirements for local health workers, significantly streamlining the program's efficiency and impact.
Case Study 2: Emergency Department Rapid Diagnosis in a European Hospital NetworkA major hospital network in Europe integrated PrisesBio's 4th Gen Rapid Test Kits into their emergency departments to provide immediate HIV status for patients presenting with non-specific symptoms, or those requiring urgent surgical procedures or organ transplantation. The 15-minute turnaround time facilitated rapid clinical decision-making, ensuring patient safety and appropriate isolation protocols where necessary. This rapid diagnostic capability minimized delays in critical care pathways, proving particularly useful for high-volume urban settings. The hospital reported a significant reduction in the average time to HIV diagnosis for new presenters, from several hours (with lab-based tests) to less than 30 minutes, drastically improving patient flow and risk management.
These cases demonstrate the practical, real-world utility and positive impact of reliable 4th generation rapid diagnostic kits in diverse healthcare environments.

Authoritativeness and Trustworthiness
Establishing trust and demonstrating authority are critical for B2B partners in the medical diagnostics industry. Manufacturers of the hiv ab/ag gen blood rapid test kit achieve this through rigorous quality systems, extensive validation, and comprehensive customer support.
Certifications and Compliance:
- ISO 13485:2016 Certification: This international standard specifies requirements for a quality management system where an organization needs to demonstrate its ability to provide medical devices and related services that consistently meet customer and applicable regulatory requirements. PrisesBio adheres strictly to this, ensuring robust design, development, production, installation, and servicing of medical devices.
- CE Mark: All kits are CE marked, indicating conformity with European Union safety, health, and environmental protection directives. This allows for free movement of the product within the European Economic Area.
- FDA (as applicable): For markets requiring specific US FDA approval, products undergo rigorous review and validation to meet their stringent standards, often including clinical trials data.
Customer Support and Warranty:
- Warranty Commitment: A comprehensive warranty is provided, typically covering manufacturing defects for the entire shelf life of the product, contingent on proper storage and handling as per instructions.
- Lead Time & Fulfillment: Standard lead times for bulk orders are typically 2-4 weeks, with expedited options available for urgent requirements, supported by a robust global logistics network.
- Dedicated Technical Support: A team of experienced product specialists and scientific experts is available to provide technical assistance, troubleshooting, and training, ensuring optimal use and performance of the kits.
Frequently Asked Questions (FAQ):
- Q1: What is the primary advantage of a 4th generation HIV rapid test over older generations?
- A1: Fourth-generation tests offer a significantly shorter diagnostic window period by simultaneously detecting both the HIV-1 p24 antigen (which appears early in infection) and antibodies to HIV-1 and HIV-2, allowing for earlier detection of acute infections.
- Q2: What sample types can be used with this kit?
- A2: The PrisesBio HIV Ab/Ag 4th Gen Blood Rapid Test Kit is validated for use with human whole blood, serum, or plasma specimens.
- Q3: How long does it take to get results?
- A3: Results are typically available within 15 to 20 minutes, making it ideal for point-of-care settings.
- Q4: Are these kits reliable for initial screening?
- A4: Yes, with high sensitivity and specificity (>99% for both), these kits are highly reliable for initial screening. Positive results should always be confirmed by a more specific, confirmatory test as per national guidelines.
- Q5: What is the shelf life and storage condition for the kit?
- A5: The kit has a shelf life of 24 months from the manufacturing date and should be stored at 2-30°C (36-86°F) to maintain optimal performance.
Conclusion
The hiv ab/ag gen blood rapid test kit represents a critical leap forward in HIV diagnostics. By simultaneously targeting both p24 antigen and HIV antibodies, these 4th generation rapid tests offer an unprecedented window for early detection, significantly reducing the time from exposure to diagnosis. This capability is instrumental in curbing the spread of HIV, facilitating prompt linkage to care, and ultimately improving patient outcomes globally. With robust technical specifications, a meticulously controlled manufacturing process, extensive application versatility, and unwavering commitment to quality and customer support, these kits are poised to continue playing a pivotal role in global health strategies, empowering healthcare providers with accurate, accessible, and rapid diagnostic solutions.
References
- World Health Organization. "Guidelines for HIV testing services: consolidated guidelines." WHO Press, 2015.
- Pauza, C. D., & HIV-1 p24 antigen. In Encyclopedia of AIDS (pp. 583-585). Springer, New York, NY.
- Centers for Disease Control and Prevention. "Laboratory Testing for the Diagnosis of HIV Infection: Updated Recommendations." CDC, 2014.
- UNAIDS. "Fast-Track Ending the AIDS Epidemic." UNAIDS, 2014.
- ISO 13485:2016 Medical devices – Quality management systems – Requirements for regulatory purposes. International Organization for Standardization.
-
Buy at home covid test rapid covid test kit antigen test covid19
NewsApr.16,2026
-
Covid19 Antigent Test Kit Cassette Covid Test Rapid Diagnostic Covid 19 Antigen Rapid Test
NewsApr.09,2026
-
Comprehensive Guide to Antigen Covid Test Covid Saliva Test and Covid Test Kit 19 Options
NewsApr.07,2026
-
Comprehensive Guide to Test Kit Covid Covid19 Rapid Antigen Test Self Test Covid Options
NewsApr.04,2026
-
Comprehensive Guide to Rapid Test Kits COVID19 for Accurate Covid Self Test Results
NewsMar.31,2026
-
Comprehensive Guide to Rapid Antigen Test Kit Covid 19 Covid Rapid Test Kit Test Kit Covid 19
NewsMar.28,2026

