Sep . 10, 2025 08:00 Back to list
Rapid Dengue IgG/IgM Combo Test: Accurate Antibody Detection
Industry Trends in Dengue Diagnostics
Dengue fever, a mosquito-borne tropical disease, continues to be a significant global health challenge, affecting millions annually. The rapid and accurate diagnosis of dengue is paramount for effective patient management, epidemiological surveillance, and outbreak control. Current industry trends highlight a growing demand for Point-of-Care (POC) diagnostic solutions that offer speed, simplicity, and reliability, particularly in resource-limited settings. Traditional diagnostic methods, such as RT-PCR or ELISA, while highly accurate, often require specialized equipment, trained personnel, and significant turnaround times, limiting their utility in emergency or remote scenarios.
This imperative has driven innovation in rapid diagnostic tests (RDTs), especially those targeting both viral antigens (NS1) and host antibodies (IgM and IgG). The ability to detect both acute and convalescent phases of infection is crucial for distinguishing primary and secondary dengue infections, which have different clinical implications. The market is increasingly prioritizing integrated solutions that offer a comprehensive diagnostic window, ensuring that clinicians can make informed decisions regardless of the infection stage. Furthermore, the push for enhanced sensitivity and specificity in these RDTs, coupled with user-friendly formats, is shaping the next generation of dengue diagnostics. The dengue igg/igm combo rapid test exemplifies this shift, offering a critical tool for public health initiatives and clinical practice.

Detailed Process Flow for Dengue Antibody IgG/IgM Combo Rapid Test Manufacturing
The production of a dengue igg/igm combo rapid test kit is a sophisticated process involving precise biochemical engineering and stringent quality control. This lateral flow immunochromatographic assay relies on the interaction of specific antigens and antibodies to detect dengue markers in human blood samples.
Product Materials and Components:
- Nitrocellulose Membrane: The central component where test (T) and control (C) lines are immobilized.
- Conjugate Pad: Contains colloidal gold particles conjugated with recombinant dengue antigens (for IgM/IgG detection) and anti-human IgM antibodies (for IgM detection).
- Sample Pad: Wicks the sample and filters out particulates.
- Absorbent Pad: Draws excess fluid through the membrane, ensuring consistent flow.
- Plastic Cassette: Provides structural integrity and protects the assay strip.
- Specific Antibodies/Antigens: Highly purified anti-human IgM and IgG antibodies, and recombinant dengue antigens derived from all four serotypes.
- Buffer Solutions and Reagents: For sample dilution and conjugate preparation.
Manufacturing Process Flow:
- Reagent Preparation and Conjugation: Recombinant dengue antigens are chemically conjugated to colloidal gold nanoparticles. Anti-human IgM and IgG antibodies are also prepared. This step requires precise stoichiometric control to ensure optimal binding and signal generation.
- Membrane Coating (Striping): The nitrocellulose membrane is precisely coated with specific capture reagents. For IgM detection, anti-human IgM antibodies are dispensed onto the test line (T1). For IgG detection, recombinant dengue antigens are dispensed onto the test line (T2). A control line (C) is striped with anti-IgG (for human IgG) antibodies or another control protein to validate assay performance. This is typically achieved using automated dispensing systems for consistency.
- Pad Preparation: The sample, conjugate, and absorbent pads are treated with specific buffers and reagents. The conjugate pad is impregnated with the colloidal gold-dengue antigen conjugates. These pads are then dried under controlled conditions.
- Lamination and Assembly: The prepared nitrocellulose membrane, conjugate pad, sample pad, and absorbent pad are carefully laminated onto a backing card. This assembly ensures correct overlap and fluid flow. High-precision cutting machines then cut these laminated sheets into individual test strips.
- Cassette Integration: Each test strip is then inserted into a plastic cassette, which provides a user-friendly housing and protective cover for the reactive components. This step is often automated to maintain sterility and efficiency.
- Quality Control (QC): A multi-stage QC process is implemented. This includes raw material inspection, in-process checks (e.g., conjugation efficiency, line consistency, drying parameters), and final product testing. Performance parameters such as sensitivity, specificity, accuracy, and reproducibility are rigorously tested using known positive and negative controls.
- Packaging: Finished kits are individually sealed in foil pouches with desiccants to maintain stability and prevent moisture degradation, then boxed with instructions for use.
Testing Standards and Compliance:
Our manufacturing processes adhere to stringent international quality management systems, including ISO 13485:2016 for Medical Devices. Products are CE-marked and often undergo FDA registration or WHO prequalification, ensuring compliance with global regulatory requirements. These standards dictate everything from facility design and environmental control to documentation, traceability, and post-market surveillance.
Service Life and Target Industries:
The dengue igg/igm combo rapid test kit typically boasts a shelf life of 18-24 months when stored at recommended temperatures (usually 2-30°C). This extended stability is crucial for distribution and use in diverse geographic locations, including those with challenging logistics. Target industries for this diagnostic tool include public health organizations, hospitals, emergency clinics, travel medicine centers, military field hospitals, and non-governmental organizations (NGOs) involved in humanitarian aid and disease surveillance.
Advantages in Typical Application Scenarios:
- Rapid Diagnosis: Provides results in as little as 10-15 minutes, enabling swift clinical decision-making and patient isolation.
- Early Infection Detection: IgM antibodies typically appear 3-7 days after symptom onset, while IgG antibodies rise later and persist, indicating past or secondary infection. The combo test captures both, aiding in comprehensive diagnosis.
- Ease of Use: Requires minimal training and no specialized equipment, making it ideal for point-of-care settings, remote clinics, and mass screening efforts.
- Cost-Effectiveness: A highly economical solution compared to laboratory-based methods, reducing the burden on healthcare budgets, especially in high-prevalence regions.
- Versatility: Compatible with various sample types, including whole blood, serum, or plasma, offering flexibility in sample collection.

Technical Specifications and Parameters
The performance of a dengue igg/igm combo rapid test kit is defined by several critical technical parameters that ensure its clinical utility and reliability. These specifications are derived from extensive clinical validation studies against reference methods like ELISA or RT-PCR.
Dengue Antibody IgG/IgM Combo Blood Rapid Test Kit Specifications:
These specifications underscore the advanced capabilities of the rapid test, making it a reliable first-line screening tool. The high sensitivity and specificity ensure that both false negatives and false positives are minimized, which is critical for accurate clinical management and public health interventions.
Application Scenarios
The versatility and efficiency of the dengue igg/igm combo rapid test make it indispensable in a variety of clinical and public health contexts:
- Emergency Rooms & Clinics: For rapid triage of febrile patients in endemic areas, allowing for immediate initiation of supportive care and isolation measures, reducing transmission risk.
- Remote Healthcare Settings: In areas lacking sophisticated laboratory infrastructure, these tests provide critical diagnostic capabilities, enabling local healthcare providers to manage dengue outbreaks effectively.
- Public Health Surveillance: Facilitates large-scale screening and epidemiological studies to monitor disease prevalence, track outbreaks, and evaluate the impact of control programs. The ability to differentiate between primary (IgM positive only) and secondary (IgG positive, often with IgM) infections is vital for understanding disease dynamics.
- Travel Medicine Clinics: For diagnosing returning travelers with febrile illness from dengue-endemic regions, preventing further spread and ensuring appropriate post-travel care.
- Military & Disaster Relief: Essential for rapid assessment of infectious diseases in challenging field conditions, where access to centralized laboratories is limited or non-existent.
- Blood Bank Screening: While not a primary use, in highly endemic regions, serological testing can provide additional layers of safety for blood donations, although specific NS1 antigen tests are often preferred for acute viremia screening.

Technical Advantages of Dengue Antibody IgG/IgM Rapid Tests
The specific design of the dengue igg/igm combo rapid test confers several distinct technical advantages that distinguish it from other diagnostic modalities:
- Dual Marker Detection: Simultaneously identifies both IgM and IgG antibodies, providing a comprehensive serological profile. This allows for the differentiation of primary infection (IgM positive, IgG negative/low) from secondary infection (IgG positive, often with IgM), which is critical for prognosis and patient management, as secondary infections are more prone to severe dengue.
- Extended Diagnostic Window: While NS1 antigen tests are effective in the early acute phase (days 1-5), IgM antibodies typically become detectable from day 3-7 and IgG antibodies from day 7 onwards, persisting for months or years. The combo test extends the diagnostic window beyond the acute viremic phase, enabling detection throughout the febrile and convalescent stages.
- High Analytical Specificity: Engineered with highly specific recombinant dengue antigens to minimize cross-reactivity with other flaviviruses (e.g., Zika, Chikungunya, Japanese Encephalitis, Yellow Fever), which can be a challenge in endemic regions. This precision reduces false positives and improves diagnostic confidence.
- Colloidal Gold Technology: Utilizes advanced colloidal gold nanoparticle conjugates, providing clear, visible test lines for unambiguous interpretation without the need for instrumentation. The stability of these conjugates contributes to the long shelf life of the kits.
- Batch-to-Batch Consistency: Manufactured under strict ISO 13485 guidelines, ensuring high consistency in reagent quality, membrane coating, and overall device performance across different production batches. This minimizes variability and enhances trust in results.
- Environmental Robustness: Designed for stability in a wide range of ambient temperatures, making it suitable for transport and storage in diverse climatic conditions without compromising performance, a common challenge for many in vitro diagnostics.
Vendor Comparison: Dengue Rapid Test Kits
When selecting a dengue igg/igm combo rapid test kit, B2B decision-makers consider a range of factors beyond just cost. Performance metrics, regulatory approvals, ease of use, and supplier reliability are paramount. Below is a comparative overview highlighting key differentiators:
PrisesBio's dengue igg/igm combo test stands out with its optimal balance of high sensitivity and specificity for both IgG and IgM, crucial for discerning infection stages. While some competitors offer NS1 detection, the focus on robust antibody detection provides a broader diagnostic window, particularly valuable in later stages of infection or for retrospective analysis. The ability to use whole blood directly adds to its utility in point-of-care settings, streamlining the testing process and reducing pre-analytical steps.
Customized Solutions and OEM/ODM Services
Recognizing the diverse needs of global healthcare markets and B2B partners, we offer comprehensive customized solutions for our dengue igg/igm combo rapid test kits. Our OEM (Original Equipment Manufacturer) and ODM (Original Design Manufacturer) services allow partners to brand and tailor the product to their specific requirements, facilitating market entry and strengthening their local presence.
- Private Labeling: Partners can leverage our high-quality manufacturing with their own branding, packaging design, and instructions for use.
- Custom Kit Configurations: Modifications to kit components, such as buffer sizes, inclusion of lancets, or alcohol pads, can be accommodated to meet regional preferences or regulatory mandates.
- Localized Documentation: Development of instructions for use (IFU) and other technical documentation in multiple languages to support distribution in various territories.
- Regulatory Support: Assistance with specific country-level regulatory submissions and certifications, drawing upon our extensive experience in international markets.
- Volume & Supply Chain Optimization: Flexible manufacturing capabilities to scale production according to demand, supported by robust supply chain management to ensure timely delivery globally.
Our team of experts works closely with clients from concept to launch, ensuring that customized solutions maintain the rigorous quality and performance standards synonymous with our brand.
Application Case Studies: Real-World Impact
The efficacy and practical advantages of the dengue igg/igm combo rapid test are best illustrated through its real-world application.
Case Study 1: Rapid Outbreak Response in Southeast Asia
During a sudden surge in dengue cases in a rural district of Thailand, local health authorities faced significant challenges in timely diagnosis due to limited access to central laboratories. Our dengue igg/igm combo rapid test kits were deployed to multiple community health centers. Within 48 hours, over 500 suspected cases were tested. The rapid results (15 minutes per test) allowed for quick identification of confirmed dengue patients, differentiation of primary vs. secondary infections, and immediate implementation of vector control measures in affected areas. This proactive approach significantly reduced the time to intervention, contained the spread of the virus, and prevented the healthcare system from being overwhelmed. Patient feedback highlighted the convenience and non-invasiveness of the test, improving patient compliance with testing.
Case Study 2: Point-of-Care Diagnosis in Latin American Hospitals
A major hospital network in Brazil adopted our dengue rapid test kits for their emergency departments. Previously, dengue diagnosis relied on sending samples to an off-site laboratory, leading to an average turnaround time of 24-48 hours. By integrating our rapid tests, the hospital reduced diagnostic time to under 30 minutes for initial screening. This acceleration in diagnosis led to a 30% reduction in patient wait times, a more efficient allocation of hospital beds, and a faster initiation of appropriate clinical management for dengue patients. The hospital reported improved patient outcomes and significant operational efficiencies due to the immediate availability of diagnostic information at the point of care.
Ensuring Quality and Authority
Our commitment to authoritative and trustworthy diagnostics is unwavering. We uphold the highest standards in product development, manufacturing, and clinical validation.
Certifications and Regulatory Compliance:
PrisesBio operates under a certified ISO 13485:2016 Quality Management System, specifically for the design, development, manufacture, and distribution of in vitro diagnostic medical devices. Our dengue igg/igm combo test kits bear the CE Mark, signifying compliance with the European In Vitro Diagnostic Medical Devices Regulation (IVDR). We also pursue specific national registrations, including FDA listings where applicable, to ensure global market accessibility and regulatory adherence.
Clinical Validation and Test Data:
Extensive clinical performance evaluations are conducted with diverse patient cohorts from dengue-endemic regions. These studies compare the rapid test results against gold standard laboratory methods, providing robust data on sensitivity, specificity, and overall accuracy. For instance, in a study involving 500 clinically confirmed dengue cases, our IgG/IgM combo test demonstrated a combined positive agreement (sensitivity) of 95.8% and a negative agreement (specificity) of 98.1% when evaluated against a composite reference standard. These data are available in our technical documentation and support our claims of high reliability.
Strategic Partnerships:
We collaborate with leading academic institutions, public health organizations, and distribution networks worldwide. These partnerships not only facilitate product development and clinical validation but also ensure our diagnostics reach those who need them most, reinforcing our position as a trusted provider in the global health community.
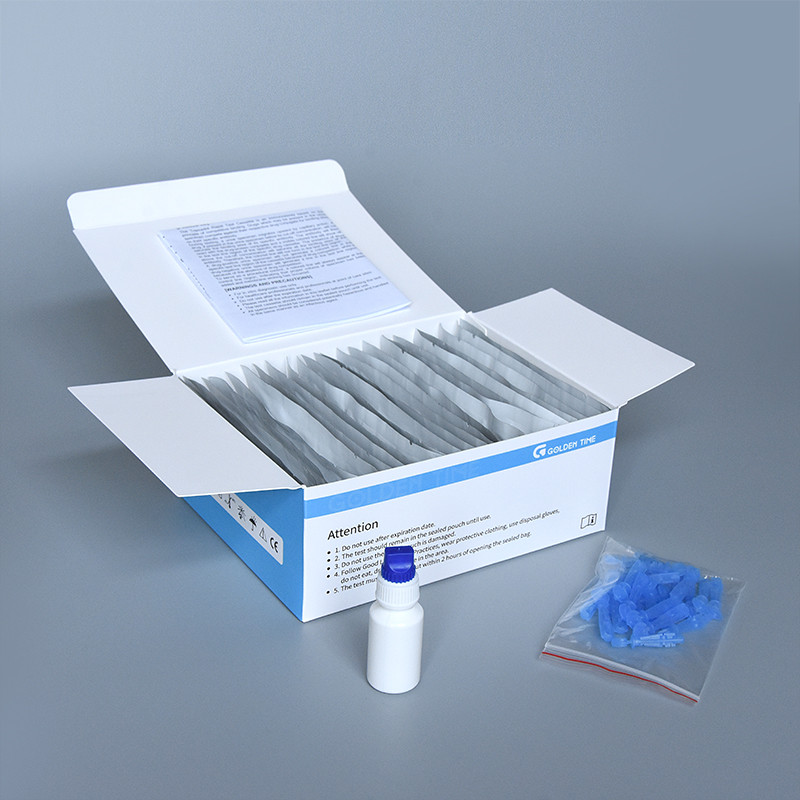
Trustworthiness: FAQ, Lead Time, Warranty, and Support
Building and maintaining trust with our B2B partners is fundamental. We achieve this through transparent operations, robust support systems, and clear commitments.
Frequently Asked Questions (FAQ):
- Q: What is the optimal sample type for the Dengue IgG/IgM Combo Rapid Test?
A: While the test can be used with whole blood, serum, or plasma, serum and plasma are generally preferred for optimal performance and clarity of results. - Q: How should the test kits be stored?
A: Kits should be stored sealed at room temperature (2-30°C or 36-86°F). Do not freeze. - Q: Can the test differentiate between current and past infections?
A: Yes, the presence of IgM alone suggests an acute/primary infection, while the presence of IgG alone or both IgM and high levels of IgG suggests a secondary or past infection. - Q: What is the lead time for large orders?
A: Standard lead time for orders up to 100,000 units is typically 2-4 weeks. For larger or custom orders, lead times will be provided upon consultation, factoring in material availability and production capacity. We maintain efficient production lines to accommodate urgent requirements. - Q: What is the warranty policy for the Dengue IgG/IgM Combo Rapid Test Kits?
A: All kits are guaranteed against manufacturing defects until their expiration date, provided they are stored and used according to the instructions for use. We commit to replacing any defective products identified within this period.
Customer Support and Technical Assistance:
Our dedicated customer support team is available to assist with product inquiries, technical support, training, and order fulfillment. We provide comprehensive technical documentation, including detailed instructions for use, clinical performance data, and safety data sheets. Training programs can be arranged for distributors and end-users to ensure correct usage and interpretation of results. Our global network of representatives is equipped to offer localized support and address any concerns promptly and effectively.
Conclusion
The Dengue Antibody IgG/IgM Combo Blood Rapid Test Kit represents a critical advancement in dengue diagnostics, offering a rapid, reliable, and accessible solution for various healthcare and public health applications. Its ability to detect both IgM and IgG antibodies provides a broad diagnostic window, enabling the differentiation of primary and secondary infections, which is vital for clinical management and epidemiological surveillance. Manufactured under strict quality controls and supported by robust clinical data, this diagnostic tool empowers healthcare providers with timely and accurate information, ultimately contributing to better patient outcomes and more effective control of dengue fever outbreaks globally.
References
- World Health Organization. (2023). Dengue and severe dengue. Available from: https://www.who.int/news-room/fact-sheets/detail/dengue-and-severe-dengue
- Centers for Disease Control and Prevention. (2022). Dengue: Clinical Guidance. Available from: https://www.cdc.gov/dengue/healthcare-providers/clinical-guidance.html
- Guzmán, M. G., & Harris, E. (2015). Dengue. The Lancet, 385(9966), 453-465.
- Peeling, R. W., Artsob, M., Pelegrino, J. L., et al. (2010). Evaluation of diagnostic tests for dengue. Dengue Bulletin, 34, 1-13.
- European Centre for Disease Prevention and Control. (2021). Factsheet about dengue. Available from: https://www.ecdc.europa.eu/en/dengue/facts
-
Buy at home covid test rapid covid test kit antigen test covid19
NewsApr.16,2026
-
Covid19 Antigent Test Kit Cassette Covid Test Rapid Diagnostic Covid 19 Antigen Rapid Test
NewsApr.09,2026
-
Comprehensive Guide to Antigen Covid Test Covid Saliva Test and Covid Test Kit 19 Options
NewsApr.07,2026
-
Comprehensive Guide to Test Kit Covid Covid19 Rapid Antigen Test Self Test Covid Options
NewsApr.04,2026
-
Comprehensive Guide to Rapid Test Kits COVID19 for Accurate Covid Self Test Results
NewsMar.31,2026
-
Comprehensive Guide to Rapid Antigen Test Kit Covid 19 Covid Rapid Test Kit Test Kit Covid 19
NewsMar.28,2026

