Aug . 31, 2025 00:40 Back to list
Accurate COVID-19 Rapid Test Kits for Fast Antigen Detection
Industry Trends & The Evolution of Rapid Diagnostic Technologies
The global landscape of infectious disease diagnostics has undergone a significant transformation, particularly in response to the COVID-19 pandemic. The demand for rapid, accurate, and accessible diagnostic tools surged, accelerating innovation in Point-of-Care (POC) testing. Before 2020, PCR (Polymerase Chain Reaction) tests were the gold standard due to their high sensitivity and specificity. However, their reliance on specialized laboratory equipment, trained personnel, and longer turnaround times posed significant challenges for widespread, immediate screening.
The advent of antigen-based rapid diagnostic tests (RDTs), specifically the covid-19 rapid test kit, marked a pivotal shift. These kits offer a decentralized testing solution, enabling quick identification of infected individuals outside of traditional laboratory settings. Market analysis indicates a consistent growth trajectory for the in-vitro diagnostics (IVD) market, with a significant segment dedicated to infectious disease diagnostics. Projections suggest a compound annual growth rate (CAGR) driven by factors such as increasing global health awareness, the emergence of novel pathogens, and advancements in biosensor technologies. This expansion is further fueled by the integration of AI and machine learning into diagnostic platforms for enhanced accuracy and predictive capabilities, although rapid antigen tests largely remain foundational for initial screening due to their simplicity and speed.
The emphasis has shifted from purely clinical laboratory testing to integrated diagnostic ecosystems that include POC devices, digital health platforms, and robust supply chain management. This ensures that essential diagnostic tools, like the covid-19 rapid test kit, are not only effective but also readily available to meet fluctuating public health demands.
Manufacturing Process Flow of a COVID-19 Rapid Test Kit
The production of a covid-19 rapid test kit, specifically a COVID-19 (SARS-CoV-2) Antigen Diagnostic Test Kit, involves a highly controlled, multi-stage manufacturing process to ensure consistent quality and performance. This process adheres strictly to international quality management systems such as ISO 13485 for medical devices, guaranteeing reliability and regulatory compliance.
Schematic Steps in Kit Production:
-
Antigen Conjugate Pad Preparation:
High-purity colloidal gold nanoparticles are conjugated with anti-SARS-CoV-2 antibodies. This solution is then precisely dispensed and dried onto a non-woven fabric pad (conjugate pad). Materials are sourced for high binding efficiency and stability, often involving automated dispensing systems for consistency.
-
Nitrocellulose Membrane Stripping:
A nitrocellulose membrane, the core of the lateral flow assay, is coated with specific capture antibodies for SARS-CoV-2 (test line, 'T') and an anti-immunoglobulin or control protein (control line, 'C'). Precision coating techniques, often employing robotic dispensers, ensure uniform line width and antigen/antibody deposition. Post-coating, membranes undergo controlled drying.
-
Sample Pad & Absorbent Pad Preparation:
A sample pad, typically made of cellulose or glass fiber, is treated to facilitate consistent sample flow and remove interfering substances. An absorbent pad, usually high-purity cellulose, is placed at the distal end of the strip to wick away excess sample and maintain a steady flow rate. Both are cut using precise CNC machining to exact dimensions.
-
Strip Assembly (Casting & Lamination):
The prepared sample pad, conjugate pad, nitrocellulose membrane, and absorbent pad are laminated onto a plastic backing card, ensuring proper overlap and adhesion. This casting process requires precision engineering to prevent misalignment, which could compromise test accuracy. Automated lamination systems maintain consistent pressure and temperature.
-
Strip Cutting & Housing Assembly:
The laminated cards are then cut into individual test strips using high-precision slitting machines. Each strip is then inserted into a plastic cassette (housing), which is typically molded through injection casting processes from medical-grade polypropylene. This protects the strip and provides a user-friendly interface.
-
Quality Control & Packaging:
Each batch undergoes rigorous quality control testing, including sensitivity, specificity, stability, and reproducibility checks against established standards (e.g., WHO emergency use listing criteria, FDA EUA guidelines). Only kits meeting ANSI/CLSI standards for diagnostic performance proceed to packaging. Kits are then individually sealed in foil pouches with desiccant to ensure a typical service life (shelf life) of 18-24 months when stored correctly (e.g., 2-30°C).
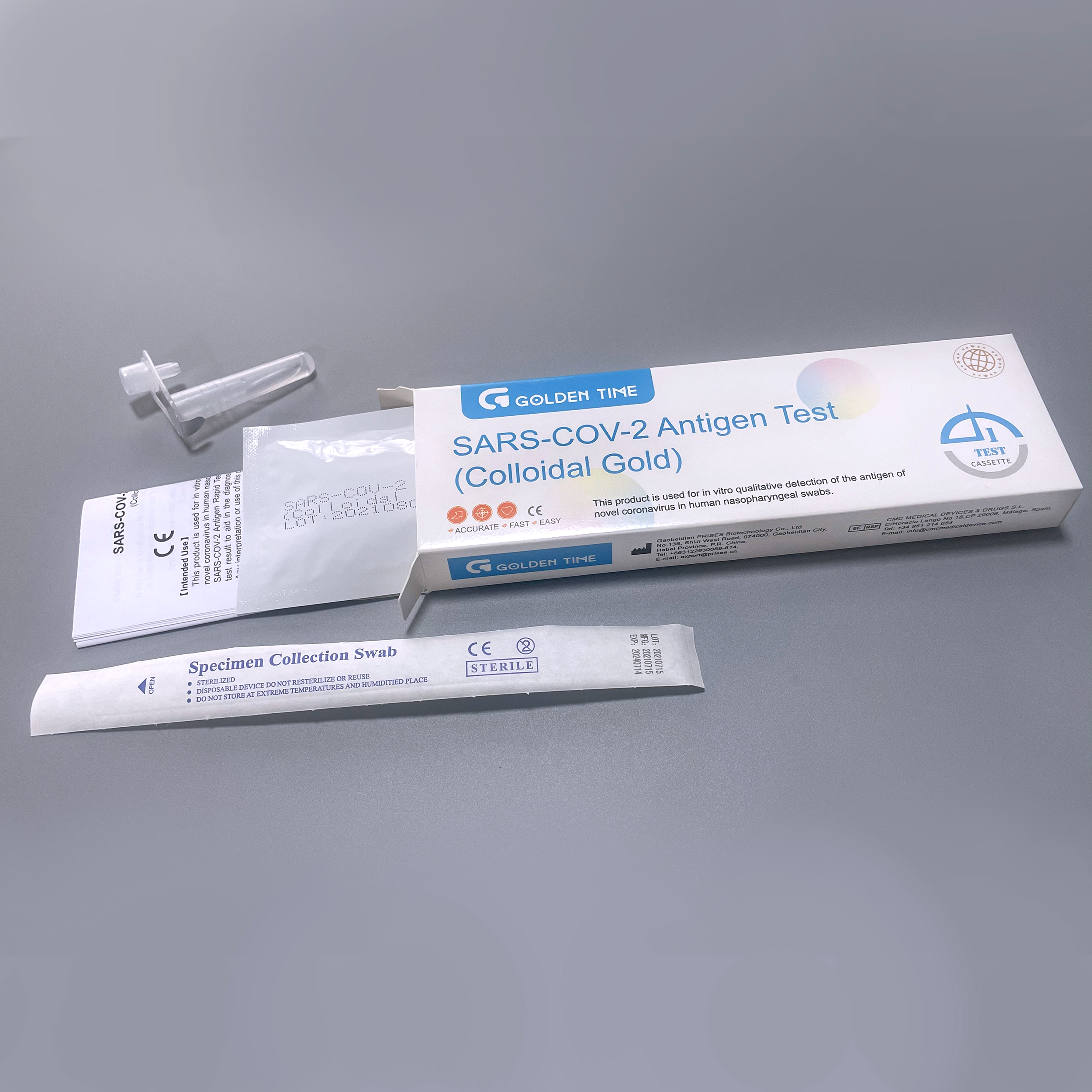
Target Industries & Advantages:
These advanced covid-19 antigen diagnostic test kit solutions are critical for diverse sectors:
- Healthcare & Public Health: Rapid screening in clinics, hospitals, and pop-up testing sites. Advantages include immediate patient triage and infection control, reducing transmission risks.
- Corporate & Industrial: Employee screening programs to maintain operational continuity. Benefits include swift identification of positive cases, enabling rapid isolation and minimizing disruption.
- Education: Regular testing in schools and universities to ensure a safer learning environment.
- Travel & Hospitality: Entry/exit screening at airports, cruise terminals, and hotels. Enables efficient health checks, fostering consumer confidence.
- Events & Entertainment: Screening attendees for large gatherings. Facilitates safer events and adherence to public health guidelines.
The primary advantage across these scenarios is rapid detection, often yielding results within 15-20 minutes, allowing for immediate action and robust epidemiological control, thereby supporting energy-saving efforts by preventing widespread outbreaks that would require more extensive, resource-intensive interventions.
Technical Specifications of the COVID-19 (SARS-CoV-2) Antigen Diagnostic Test Kit
Understanding the precise technical parameters of a covid-19 rapid test kit is crucial for B2B decision-makers assessing its suitability for various deployment scenarios. Our COVID-19 (SARS-CoV-2) Antigen Diagnostic Test Kit is engineered for high performance and reliability.
Key Performance Characteristics:
These specifications underline the kit's robust diagnostic capabilities, providing confidence to professionals requiring an efficient and reliable covid-19 antigen diagnostic test kit for mass screening or individual assessment. The high sensitivity ensures minimal false negatives, critical for public health, while the high specificity minimizes false positives, reducing unnecessary follow-up actions and anxiety.
Versatile Application Scenarios
The adaptability of the covid-19 rapid test kit makes it an invaluable tool across a multitude of environments where swift diagnostic results are paramount. Its ease of use and rapid turnaround time are particularly beneficial in settings that require immediate decision-making regarding isolation, contact tracing, and public health interventions.
Key Application Environments:
-
Corporate Wellness & Return-to-Office Programs:
Companies globally have implemented routine testing protocols using rapid antigen kits to ensure a safe working environment. This minimizes business disruption and fosters employee confidence, particularly in industries like manufacturing, logistics, and professional services where on-site presence is crucial.
-
Educational Institutions:
Schools, colleges, and universities utilize these kits for regular student and staff screening. This strategy helps to quickly identify and isolate positive cases, preventing widespread outbreaks within academic communities and maintaining continuity of learning.
-
Travel & Border Control:
Airports, seaports, and border crossings deploy rapid testing for incoming and outgoing travelers. The quick results facilitate efficient passenger processing while mitigating the risk of international transmission, supporting global economic recovery.
-
Healthcare & Long-Term Care Facilities:
Beyond initial diagnosis, these kits are used for rapid screening of patients, visitors, and healthcare workers to prevent nosocomial infections and protect vulnerable populations. They enable faster triaging in emergency departments.
-
Mass Gatherings & Events:
For concerts, sports events, and conferences, rapid testing serves as an essential entry requirement, providing an additional layer of safety for attendees and organizers. This supports the safe resumption of large-scale public events.
-
Remote and Underserved Areas:
Due to their minimal infrastructure requirements, rapid kits are ideal for deployment in areas with limited access to centralized laboratories, ensuring equitable access to diagnostic testing.
In each of these scenarios, the covid-19 antigen diagnostic test kit provides a practical, scalable, and effective solution for immediate infection status determination, playing a crucial role in managing public health challenges.
Technical Advantages & Vendor Comparison
The technical advantages of a high-quality covid-19 rapid test kit are multifaceted, positioning it as a preferred diagnostic tool for immediate screening and surveillance. These advantages translate directly into operational efficiencies and enhanced public health outcomes for our B2B clients.
Core Technical Advantages:
- Rapid Turnaround Time: Delivers results in 15-20 minutes, significantly faster than PCR tests (hours to days), enabling immediate isolation and contact tracing.
- Ease of Use: Designed for simplicity, requiring minimal training. This facilitates decentralized testing by non-laboratory personnel in various settings.
- Cost-Effectiveness: Significantly more affordable per test than PCR, making large-scale and frequent screening economically viable.
- Portability & No Specialized Equipment: Operates without electricity or complex laboratory instruments, making it ideal for remote locations and point-of-care use.
- High Specificity: Minimizes false positives, which is crucial for preventing unnecessary isolation and follow-up tests, optimizing resource allocation.
- Scalability: Manufacturing processes are highly scalable, allowing for rapid production to meet global demand surges.

Vendor Comparison Table: Key Differentiators for COVID-19 Antigen Test Kits
When selecting a covid-19 antigen diagnostic test kit, discerning buyers analyze factors beyond basic performance. This comparison highlights common differentiators:
Our commitment to superior performance and comprehensive regulatory compliance positions our COVID-19 (SARS-CoV-2) Antigen Diagnostic Test Kit as a leading solution in the market, providing unmatched reliability and ease of integration for demanding B2B applications.
Customized Solutions & Application Case Studies
Recognizing the diverse needs of B2B clients, we offer extensive customization options for our covid-19 rapid test kit solutions. Beyond standard offerings, our capabilities extend to tailoring aspects of the product and service to perfectly align with specific operational requirements and brand identities.
Customization Capabilities:
- OEM/ODM Services: Full Original Equipment Manufacturer (OEM) and Original Design Manufacturer (ODM) capabilities allow clients to brand our diagnostic kits under their own label. This includes custom packaging design, instruction manuals, and kit component configurations.
- Packaging & Kitting: Tailored packaging solutions for bulk orders, multi-packs, or specific point-of-care distribution needs. We can also integrate additional components, such as digital result readers or tele-health support information, into custom kits.
- Regulatory Support: Assistance with specific national or regional regulatory submissions (e.g., local health authority approvals) to ensure smooth market entry and compliance for our partners.
- Logistics & Supply Chain Integration: Customized delivery schedules, warehousing, and global logistics support to ensure timely and efficient distribution, critical for large-scale procurement.
Real-World Application Case Studies:
Our covid-19 antigen diagnostic test kit has been instrumental in supporting numerous organizations globally. Below are illustrative examples of successful deployments:
Case Study 1: Large Automotive Manufacturing Plant (USA)
- Challenge: Maintain continuous production amidst potential COVID-19 outbreaks among a workforce of 5,000 employees. PCR testing turnaround times were too slow, leading to significant operational delays and potential transmission.
- Solution: Implementation of a daily rapid antigen testing program for all employees entering the facility, using our customized rapid test kits. We provided kits with integrated QR codes for digital result reporting and tailored training materials for on-site health staff.
- Outcome: Reduced average time to identify positive cases from 48 hours to 20 minutes. This led to a 70% reduction in production line stoppages due to suspected cases, maintaining critical manufacturing schedules and ensuring employee safety. The program achieved a 99% employee participation rate due to ease of use.
Case Study 2: International Cruise Line Operator (Europe)
- Challenge: Re-establish passenger confidence and comply with stringent international health regulations for cruise travel, requiring pre-boarding and on-board diagnostic capabilities.
- Solution: Supplied millions of our covid-19 antigen diagnostic test kits for passenger and crew screening across their fleet. We developed specialized multi-language packaging and provided bulk logistics solutions to ports worldwide.
- Outcome: Enabled the cruise line to safely resume operations, meeting regulatory demands with high efficiency. The rapid testing protocol, coupled with other safety measures, resulted in zero reported large-scale on-board outbreaks attributed to initial screening failures, significantly boosting consumer trust and booking rates.
These case studies highlight our capability to not only supply high-performance diagnostic products but also to partner with clients to develop integrated, tailored solutions that address complex operational and public health challenges.
Frequently Asked Questions (FAQ)
Q: What is the shelf life of the COVID-19 (SARS-CoV-2) Antigen Diagnostic Test Kit?
A: Our kits typically have a shelf life of 18-24 months from the date of manufacture when stored correctly at 2-30°C (36-86°F).
Q: Can the kit detect new SARS-CoV-2 variants?
A: Our covid-19 antigen diagnostic test kit targets the highly conserved nucleocapsid protein, which has shown minimal mutation across known major variants (e.g., Alpha, Delta, Omicron). Ongoing validation ensures continued efficacy against emerging variants.
Q: What is the minimum order quantity (MOQ) for bulk purchases?
A: MOQ varies depending on customization requirements and current stock. Please contact our sales team directly for specific details and competitive pricing for large volumes.
Q: Are training materials provided for kit usage?
A: Yes, comprehensive instructions for use (IFU) are included with each kit. For large deployments, we can provide additional digital training resources, webinars, and on-site support as part of our customized solutions package.
Lead Time, Warranty & Customer Support
Lead Time & Fulfillment:
Our production facilities are equipped for high-volume manufacturing, ensuring robust supply chain resilience. Standard orders for the covid-19 rapid test kit are typically fulfilled within 7-14 business days, depending on order size and destination. For large-scale or custom orders, lead times will be provided upon detailed quotation, factoring in specific customization requirements and logistical complexities. We maintain buffer stocks to respond swiftly to urgent demands.
Warranty Commitments:
All our COVID-19 (SARS-CoV-2) Antigen Diagnostic Test Kits are manufactured under stringent quality control (ISO 13485 certified) and are guaranteed to meet the published specifications and performance parameters until their stated expiration date, provided they are stored and used as per the instructions for use. Any defective products identified within the warranty period will be replaced or refunded. Specific terms and conditions apply and are available upon request.
Dedicated Customer Support:
We are committed to providing unparalleled support to our B2B partners. Our expert customer service team is available to assist with product inquiries, technical support, order tracking, and post-sales assistance. For immediate assistance, please contact us via:
- Email: sales@prisesbio.com
- Phone: +1 (XXX) XXX-XXXX (Available during business hours)
- Online Portal: Secure client portal for order management and documentation.
Our goal is to ensure seamless integration and optimal performance of our diagnostic solutions within your operational framework.
References
- World Health Organization. (2020). Antigen-detection in the diagnosis of SARS-CoV-2 infection. Interim Guidance.
- U.S. Food and Drug Administration. (2020). In Vitro Diagnostics EUAs - Antigen Diagnostic Tests for SARS-CoV-2.
- International Organization for Standardization. (2016). ISO 13485:2016 Medical devices – Quality management systems – Requirements for regulatory purposes.
- Clinical and Laboratory Standards Institute. (2018). CLSI EP12-A2: User Protocol for Evaluation of Qualitative Test Performance.
-
Buy at home covid test rapid covid test kit antigen test covid19
NewsApr.16,2026
-
Covid19 Antigent Test Kit Cassette Covid Test Rapid Diagnostic Covid 19 Antigen Rapid Test
NewsApr.09,2026
-
Comprehensive Guide to Antigen Covid Test Covid Saliva Test and Covid Test Kit 19 Options
NewsApr.07,2026
-
Comprehensive Guide to Test Kit Covid Covid19 Rapid Antigen Test Self Test Covid Options
NewsApr.04,2026
-
Comprehensive Guide to Rapid Test Kits COVID19 for Accurate Covid Self Test Results
NewsMar.31,2026
-
Comprehensive Guide to Rapid Antigen Test Kit Covid 19 Covid Rapid Test Kit Test Kit Covid 19
NewsMar.28,2026

