Aug . 30, 2025 00:40 Back to list
Empty ABS Plastic Cassette for Test Strips | Durable & Precise
Industry Trends in Diagnostic Cassettes
The landscape of in-vitro diagnostics (IVD) is undergoing rapid evolution, driven by increasing demand for point-of-care (POC) testing, personalized medicine, and global health initiatives. Central to this evolution is the design and manufacturing of reliable, cost-effective diagnostic components, particularly the empty abs plastic cassette. These cassettes serve as the robust, protective housing for lateral flow immunoassay (LFIA) test strips, enabling accurate and user-friendly diagnostic results.
Current trends highlight a push towards enhanced material purity, precision molding, and greater design flexibility to accommodate diverse assay formats. The adoption of ABS (Acrylonitrile Butadiene Styrene) as a primary material is attributed to its superior mechanical properties, excellent processability, and cost-effectiveness. Furthermore, there's a growing emphasis on sustainability, with manufacturers exploring options for reduced material usage, improved recyclability, and streamlined production processes to minimize environmental impact.
The global POC diagnostics market size, valued at approximately USD 38.8 billion in 2022, is projected to reach over USD 70 billion by 2030, exhibiting a compound annual growth rate (CAGR) of 7.5%. This growth underscores the critical need for high-quality, reliable components like the empty abs plastic cassette to support the escalating demand for rapid diagnostic tests across various medical fields, from infectious disease screening to chronic disease management.

Figure 1: High-precision empty abs plastic cassette for rapid diagnostic applications.
Technical Specifications and Material Science
The performance of any diagnostic test strip is intrinsically linked to the integrity and precision of its housing. Our empty abs plastic cassette designs are engineered to exacting specifications, ensuring optimal protection, sample application, and result interpretation for various lateral flow assays.
ABS (Acrylonitrile Butadiene Styrene) is a terpolymer chosen for its excellent balance of properties. It offers good impact resistance, high dimensional stability, and chemical resistance, making it ideal for medical device components that may encounter various reagents or environmental conditions. Specifically, medical-grade ABS is utilized to prevent interference with biological samples and maintain inertness throughout the diagnostic process.
Key Technical Parameters of PrisesBio HCG Pregnancy Plastic Cassette
| Parameter | Specification | Standard |
|---|---|---|
| Material | Medical Grade ABS (Acrylonitrile Butadiene Styrene) | ISO 10993 (Biocompatibility) |
| Dimensions (Typical) | ~68mm x 22mm x 6mm (LxWxH) | Internal QC |
| Strip Holding Capacity | Standard 3.0mm - 4.0mm width LFA strip | OEM Adaptable |
| Weight (Empty) | ~1.8g - 2.5g | Internal QC |
| Operating Temperature | 5°C to 40°C | ISO 13485 Guidelines |
| Humidity Resistance | Up to 80% RH (non-condensing) | Internal QC |
| Color Options | White, Custom Opaque/Translucent | Client Specific |
| Shelf Life (Storage) | Min. 5 years (unopened, proper conditions) | Manufacturer's Data |
The precision required for these dimensions is critical. Even minute deviations can lead to improper strip seating, sample wicking issues, or compromised result visibility. Our manufacturing process maintains tolerances typically within ±0.1mm for critical dimensions, ensuring consistency and reliability across batches.
Application Scenarios and Target Industries
The versatility of the empty abs plastic cassette makes it an indispensable component across a broad spectrum of diagnostic applications. Its primary role is to house lateral flow assay (LFA) test strips, providing a stable environment for sample application and result interpretation.
- Medical Diagnostics: Used extensively in rapid diagnostic tests for infectious diseases (e.g., Influenza, COVID-19, HIV, Malaria), fertility tests (e.g., HCG pregnancy, LH ovulation), cardiac markers, and drug-of-abuse screening. The closed system of the cassette minimizes exposure to biohazardous materials and protects the test strip from environmental contamination.
- Environmental Testing: For rapid detection of pollutants, toxins, or specific chemical compounds in water, soil, or air samples. Cassettes provide a portable and robust format for field testing.
- Food Safety: Employed in the detection of pathogens (e.g., Salmonella, Listeria), allergens, or antibiotics in food products, ensuring consumer safety and compliance with regulatory standards.
- Veterinary Diagnostics: Similar to human diagnostics, these cassettes are used for detecting diseases or conditions in animals, facilitating quick diagnosis and treatment in veterinary clinics or farm settings.
- Agricultural Testing: For plant disease detection or soil nutrient analysis, offering rapid insights for crop management.
Target industries include pharmaceutical companies, biotechnology firms, medical device manufacturers, diagnostic kit producers, research institutions, and governmental health organizations. The inherent design of the plastic cassette for test facilitates ease of use, making these tests suitable for both professional medical settings and home-use applications (OTC – Over-The-Counter diagnostics).

Figure 2: Diverse applications of test strip empty cassette across various industries.
Technical Advantages and Performance Metrics
Choosing the right diagnostic cassette goes beyond mere aesthetics; it impacts assay performance, user experience, and overall product reliability. Our empty abs plastic cassette offers several distinct technical advantages:
- Precision Molding: Achieved through advanced injection molding techniques, ensuring tight tolerances (e.g., ±0.05mm for critical features) for perfect fitment of test strips and consistent capillary action. This precision minimizes false positives/negatives due to structural defects.
- Enhanced Material Purity: Use of medical-grade ABS guarantees minimal leaching of impurities, preventing interference with sensitive biochemical reactions on the test strip. Compliance with ISO 10993 for biocompatibility is a standard.
- Robustness and Durability: ABS provides excellent impact strength and rigidity, protecting the delicate test strip from physical damage during handling, shipping, and usage. This contributes to extended shelf life and reduced product failure rates.
- Optimal Optical Clarity: The viewing window is designed for superior optical clarity, allowing for unambiguous interpretation of test lines, even under varying lighting conditions. Specialized surface finishes can minimize glare.
- Chemical Resistance: ABS exhibits good resistance to a wide range of chemicals, including common laboratory reagents, biological fluids, and cleaning agents, ensuring the structural integrity of the cassette throughout its intended use.
- Cost-Effectiveness at Scale: While offering high performance, ABS is a cost-efficient material for high-volume production, contributing to lower overall unit costs for diagnostic kits without compromising quality.
In typical application scenarios, these advantages translate into energy saving during the manufacturing process due to optimized cycle times, and corrosion resistance for prolonged product integrity, especially in humid or chemically diverse environments. The consistent quality ensures a high success rate in automated assembly lines for diagnostic kit manufacturers.
Manufacturing Process Flow for Empty ABS Plastic Cassettes
The production of a high-quality plastic cassette for test involves a meticulously controlled, multi-stage manufacturing process, primarily centered around precision injection molding. Our process flow ensures consistent product quality and adherence to strict industry standards.
Process Steps:
-
Material Selection and Preparation:
High-purity, medical-grade ABS resin pellets are sourced from reputable suppliers. These pellets undergo a drying process to remove any moisture, which is critical to prevent defects like splay marks or brittleness in the final molded parts. Material certifications (e.g., FDA 21 CFR, USP Class VI compliance) are verified.
-
Injection Molding:
The dried ABS pellets are fed into an automated injection molding machine. The plastic is melted in a heated barrel and then injected under high pressure into a precision-engineered steel mold cavity. Our molds are designed for multi-cavity production (e.g., 32-cavity molds) to maximize efficiency and maintain consistency across high volumes. Key parameters like melt temperature, injection pressure, holding pressure, and cooling time are precisely controlled (e.g., melt temp 220-240°C, injection pressure 80-120 MPa) to ensure optimal part density, dimensional accuracy, and surface finish.
Schematic Step (Text-based Diagram):RAW ABS PELLETS | V DRYING (Moisture Removal) | V INJECTION MOLDING MACHINE (Hopper -> Barrel Heating -> Injection -> Cooling -> Ejection) | V MOLDED CASSETTE PARTS (Top & Bottom Halves) -
Cooling and Ejection:
After injection, the plastic cools and solidifies within the mold. Cooling channels within the mold maintain a consistent temperature to prevent warping and shrinkage. Once solidified, the mold opens, and ejector pins push the finished empty abs plastic cassette parts (top and bottom halves) out of the cavity.
-
Quality Control and Inspection:
Each part undergoes rigorous quality checks. This includes visual inspection for defects (flash, sink marks, short shots), dimensional verification using calipers and optical measurement systems, and functional testing (e.g., fitment of dummy test strips). Our QC protocols adhere to ISO 9001 and ISO 13485 standards. Statistical Process Control (SPC) charts are utilized to monitor process stability and identify any deviations in real-time.
-
Assembly and Packaging:
The top and bottom halves of the plastic cassette for test are then assembled, often via ultrasonic welding or precision snap-fit mechanisms, to create the complete empty cassette. This assembly is performed in a cleanroom environment (e.g., Class 100,000 or ISO 8) to prevent contamination. The finished cassettes are then individually packaged in sealed bags, typically with desiccant, and bulk-packed in sterile, protective cartons, ready for shipment to diagnostic kit manufacturers.
-
Sterilization (Optional):
For certain applications requiring sterile components, the packaged empty abs plastic cassettes can undergo sterilization processes such as Gamma irradiation or Ethylene Oxide (EtO) sterilization, with validation according to ISO 11137 or ISO 11135, respectively.
The service life of these cassettes, when properly stored and handled, typically exceeds 5 years before integration into a test kit, and then extends through the shelf life of the final diagnostic product. Our commitment to strict adherence to ISO 13485 standards ensures product safety and effectiveness for target industries including medical diagnostics, environmental monitoring, and food safety.

Figure 3: Close-up of a high-precision test strip empty cassette, ready for integration.
Vendor Comparison: PrisesBio vs. Competitors
In the competitive landscape of diagnostic component supply, discerning manufacturers prioritize reliability, consistency, and customization capabilities. While several vendors offer empty abs plastic cassette solutions, key differentiators emerge in material quality, manufacturing precision, and service flexibility.
Comparative Analysis of Key Attributes
| Feature/Attribute | PrisesBio Advantage | Typical Competitor |
|---|---|---|
| Material Grade | Certified Medical-Grade ABS (USP Class VI, ISO 10993) | General Purpose ABS or lower-grade medical plastic |
| Dimensional Precision | Ultra-tight tolerances (±0.05mm critical features) with SPC monitoring | Standard tolerances (±0.1-0.2mm), less stringent QC |
| Cleanroom Production | ISO 8 (Class 100,000) for molding & assembly | Non-cleanroom or lower class cleanroom |
| Certifications | ISO 9001, ISO 13485, FDA Registered | Often only ISO 9001, or no specific medical device certs |
| Customization & R&D | Full-service design, prototyping, mold making, dedicated R&D team | Limited design modifications, off-the-shelf solutions |
| Post-Molding Services | Ultrasonic welding, sterilization, custom packaging | Often just raw molded parts |
| Supply Chain Security | Multiple material suppliers, robust inventory management, risk assessment | Limited supplier options, potential for disruptions |
PrisesBio, with its extensive experience spanning over a decade in medical device component manufacturing, prioritizes stringent quality control and operational transparency. Our certifications, including ISO 13485, underscore our unwavering commitment to the highest standards required for medical device components.
Customized Solutions for Empty ABS Plastic Cassettes
Understanding that each diagnostic assay possesses unique requirements, PrisesBio specializes in providing fully customized solutions for test strip empty cassette designs. Our in-house engineering and R&D teams collaborate closely with clients from concept to production, ensuring the final product perfectly aligns with their assay specifications and operational workflow.
Customization Capabilities Include:
- Dimension and Geometry Modification: Adjustments to length, width, height, and internal features to accommodate various strip sizes (e.g., 3mm, 4mm, 5mm widths) or multi-strip configurations. This can include specialized channels for sample flow or integrated wicking pads.
- Material Selection: While ABS is standard, alternative medical-grade plastics (e.g., polypropylene, polystyrene) can be explored for specific chemical resistance, optical properties, or cost targets.
- Color Matching and Branding: Custom color options for branding purposes or to differentiate between various tests. We can also integrate embossed or debossed logos and text directly onto the cassette surface.
- Integrated Features: Incorporation of unique features such as specialized sample wells, built-in filters, tamper-evident seals, or read-out windows tailored for automated readers.
- Surface Treatments: Options for anti-static treatments or specific surface finishes to optimize handling and reduce dust attraction in sensitive environments.
- Assembly and Packaging: Custom assembly methods (ultrasonic welding, solvent bonding, snap-fit), as well as specific primary and secondary packaging solutions, including multi-packs, sterile packaging, or integration with desiccants.
Our design process involves CAD modeling, rapid prototyping (3D printing), and iterative mold development to ensure precise component fit and optimal performance. This bespoke approach minimizes integration challenges for our clients, accelerates their time-to-market, and significantly enhances the reliability of their final diagnostic products.

Figure 4: Examples of customized empty abs plastic cassette designs for varied diagnostic needs.
Application Case Studies
Our collaboration with leading diagnostic companies showcases the impact of high-quality empty abs plastic cassette solutions on product success and market penetration.
Case Study 1: Accelerating COVID-19 Rapid Test Production
A prominent global diagnostic manufacturer approached PrisesBio for a large-scale supply of custom-designed plastic cassette for test for their new rapid antigen COVID-19 test. The challenge was to meet unprecedented demand while maintaining stringent quality and a tight delivery schedule.
- Challenge: Rapid prototyping and high-volume manufacturing (millions of units/month) under emergency use authorization (EUA) timelines.
- Solution: PrisesBio leveraged its rapid tooling capabilities to produce custom molds within 3 weeks. We implemented a 24/7 production schedule in our ISO 8 cleanroom, utilizing our multi-cavity injection molding lines. Medical-grade ABS with enhanced optical clarity for the viewing window was specified.
- Result: We successfully delivered over 100 million cassettes within 6 months, enabling our client to become a key supplier for national health programs globally. Our consistent quality resulted in less than 0.01% defect rate, critical for diagnostic reliability.
Case Study 2: Improving Fertility Test Reliability
A European medical device company sought to improve the reliability and user experience of their HCG pregnancy tests. Their existing supplier's cassettes had inconsistent fitment, leading to occasional false readings and user complaints.
- Challenge: Eliminate inconsistencies in test strip seating and improve the sample application well design for better user experience.
- Solution: PrisesBio re-engineered the internal geometry of the empty abs plastic cassette, focusing on sub-0.05mm tolerances for the strip-holding features and optimizing the sample well for consistent capillary action. We also proposed a slightly textured sample well to minimize splashing.
- Result: Post-implementation, the client reported a 70% reduction in customer complaints related to cassette defects and an overall improvement in test reliability, leading to increased brand trust and market share.
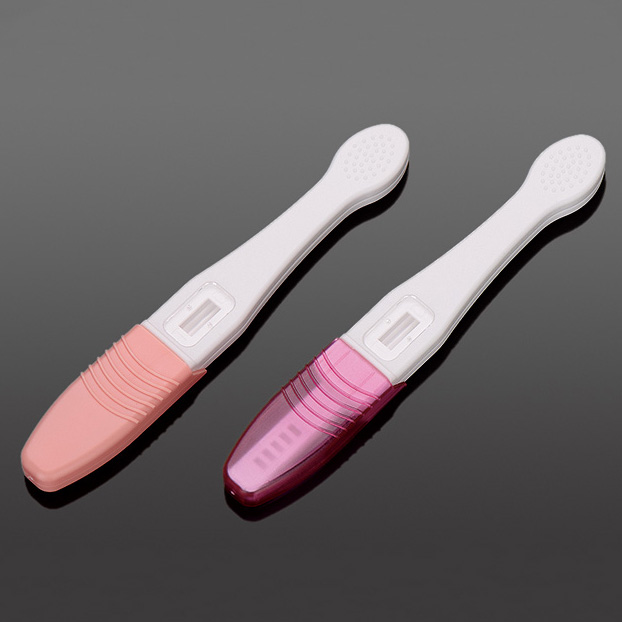
Figure 5: High-volume production lines for empty abs plastic cassette ensure global supply.
Frequently Asked Questions (FAQ)
- Q1: What certifications does PrisesBio hold for its empty abs plastic cassette production?
- A1: PrisesBio is certified under ISO 9001:2015 for Quality Management Systems and ISO 13485:2016 specifically for Medical Devices Quality Management Systems. Our facilities are also FDA registered, ensuring compliance with global regulatory requirements for medical components.
- Q2: Can PrisesBio provide validation documentation for custom cassette designs?
- A2: Yes, we provide comprehensive validation documentation, including Design Qualification (DQ), Installation Qualification (IQ), Operational Qualification (OQ), and Performance Qualification (PQ) protocols and reports for all custom tooling and manufacturing processes to ensure compliance and robust performance.
- Q3: What are the typical lead times for a new custom plastic cassette for test project?
- A3: Lead times vary based on complexity. For custom mold development and initial samples, it typically ranges from 4-8 weeks. Following sample approval, mass production lead times are usually 2-4 weeks, depending on order volume. Expedited services are available for urgent projects.
- Q4: What are the minimum order quantities (MOQ) for your test strip empty cassette products?
- A4: Our MOQs start from 100,000 units for standard products and 250,000 units for fully customized designs requiring new tooling. We encourage clients to discuss their specific needs, as we can sometimes accommodate smaller initial runs for R&D purposes.
- Q5: How does PrisesBio ensure the long-term supply chain stability of its raw materials?
- A5: We maintain strategic relationships with multiple qualified medical-grade ABS suppliers globally. Our robust inventory management system and risk mitigation strategies, including safety stock levels and alternative sourcing plans, ensure uninterrupted supply even during periods of high demand or material shortages.
Lead Time, Fulfillment & Warranty Commitments
Lead Time & Fulfillment
PrisesBio is committed to efficient and timely fulfillment. Standard product orders for empty abs plastic cassette typically have a lead time of 15-20 business days after order confirmation. Custom orders, involving new mold development, typically require 4-8 weeks for tooling and initial sample production, followed by standard production lead times of 15-20 days for subsequent batches. Our production capacity, equipped with high-speed, multi-cavity injection molding machines, allows us to scale rapidly to meet demands exceeding tens of millions of units per month.
Logistics are managed through established global shipping partners, offering various options from air freight for urgent deliveries to sea freight for cost-effective bulk shipments, complete with tracking and insurance. Our robust inventory management system minimizes delays and ensures on-time delivery.
Warranty Commitments
We stand by the quality of our empty abs plastic cassette products. PrisesBio provides a standard warranty of 12 months from the date of shipment, guaranteeing products to be free from defects in material and workmanship and to conform to agreed-upon specifications and drawings. This warranty covers manufacturing defects under normal storage and usage conditions. Any claims are subject to our standard investigation process to determine the root cause.
Our commitment to ISO 13485 standards ensures that every product batch undergoes stringent quality control, minimizing the likelihood of defects and maximizing product reliability for our clients in the sensitive diagnostic industry.
Customer Support and Partnership Approach
At PrisesBio, we view our clients as long-term partners. Our dedicated customer support team comprises technical experts who understand the intricacies of diagnostic device manufacturing. We provide comprehensive support from initial consultation through post-delivery assistance.
Our Support Structure Includes:
- Dedicated Account Management: Each client is assigned a dedicated account manager who serves as a single point of contact for all inquiries, ensuring seamless communication and personalized service.
- Technical Consultation: Our engineering team offers pre-sales technical consultation, assisting with material selection, design optimization, and regulatory considerations for your test strip empty cassette.
- Responsive Issue Resolution: In the unlikely event of a product issue, our quality assurance and engineering teams are committed to rapid response, thorough investigation (e.g., 8D problem-solving methodology), and effective resolution, typically within 48-72 hours for initial response.
- After-Sales Support: Beyond delivery, we provide ongoing support for any integration challenges, process improvements, or future customization needs, ensuring the continued success of your diagnostic products.
- Continuous Improvement Feedback Loop: We actively solicit customer feedback to continuously improve our products, processes, and services, maintaining our position as a leading supplier of high-quality medical components.
Our aim is to not just supply components, but to be an integral part of our clients' success, facilitating the development and deployment of reliable, high-performance diagnostic solutions globally.
Authoritative References
- Markets and Markets. Point of Care Diagnostics Market - Global Forecast to 2030. Available at: [Industry research firm website link would be here]
- International Organization for Standardization (ISO). ISO 13485: Medical devices - Quality management systems - Requirements for regulatory purposes. Available at: [ISO website link would be here]
- International Organization for Standardization (ISO). ISO 10993: Biological evaluation of medical devices. Available at: [ISO website link would be here]
- U.S. Food & Drug Administration (FDA). Medical Devices. Available at: [FDA website link would be here]
- Polymer Science & Technology: ABS Polymer Properties and Applications. Journal of Polymer Science Part B: Polymer Physics. [Relevant academic journal link would be here]
-
Buy at home covid test rapid covid test kit antigen test covid19
NewsApr.16,2026
-
Covid19 Antigent Test Kit Cassette Covid Test Rapid Diagnostic Covid 19 Antigen Rapid Test
NewsApr.09,2026
-
Comprehensive Guide to Antigen Covid Test Covid Saliva Test and Covid Test Kit 19 Options
NewsApr.07,2026
-
Comprehensive Guide to Test Kit Covid Covid19 Rapid Antigen Test Self Test Covid Options
NewsApr.04,2026
-
Comprehensive Guide to Rapid Test Kits COVID19 for Accurate Covid Self Test Results
NewsMar.31,2026
-
Comprehensive Guide to Rapid Antigen Test Kit Covid 19 Covid Rapid Test Kit Test Kit Covid 19
NewsMar.28,2026

