The world has faced unprecedented challenges in recent years with the emergence of infectious diseases, demanding rapid and accurate diagnostic tools. The covid rapid test has become a cornerstone of global health strategies, enabling swift detection and informed decision-making. Its significance stems not only from identifying positive cases but also in controlling outbreaks, easing burdens on healthcare systems, and facilitating a safe return to normalcy.
The increasing demand for accessible and reliable testing solutions has driven innovation in rapid diagnostics. These tests, designed for point-of-care use, offer a crucial advantage over traditional laboratory-based methods—speed. This speed translates to quicker isolation of infected individuals, prompt contact tracing, and ultimately, a reduction in community transmission.
Understanding the science, applications, and limitations of the covid rapid test is vital for healthcare professionals, policymakers, and the public alike. This knowledge empowers informed choices and strengthens our collective response to ongoing and future public health emergencies.


The covid rapid test has played an invaluable role in the global response to the pandemic. According to the World Health Organization (WHO), widespread testing is a critical component of containing outbreaks. The availability of these tests, particularly in resource-limited settings, has been instrumental in identifying and isolating cases, slowing the spread of the virus and protecting vulnerable populations.
Data from the UN indicates that rapid testing has been essential for economic recovery, allowing for the safe reopening of businesses and schools. The speed and accessibility of covid rapid tests empower individuals to take proactive steps to protect themselves and their communities, contributing to a more resilient and prepared global society.
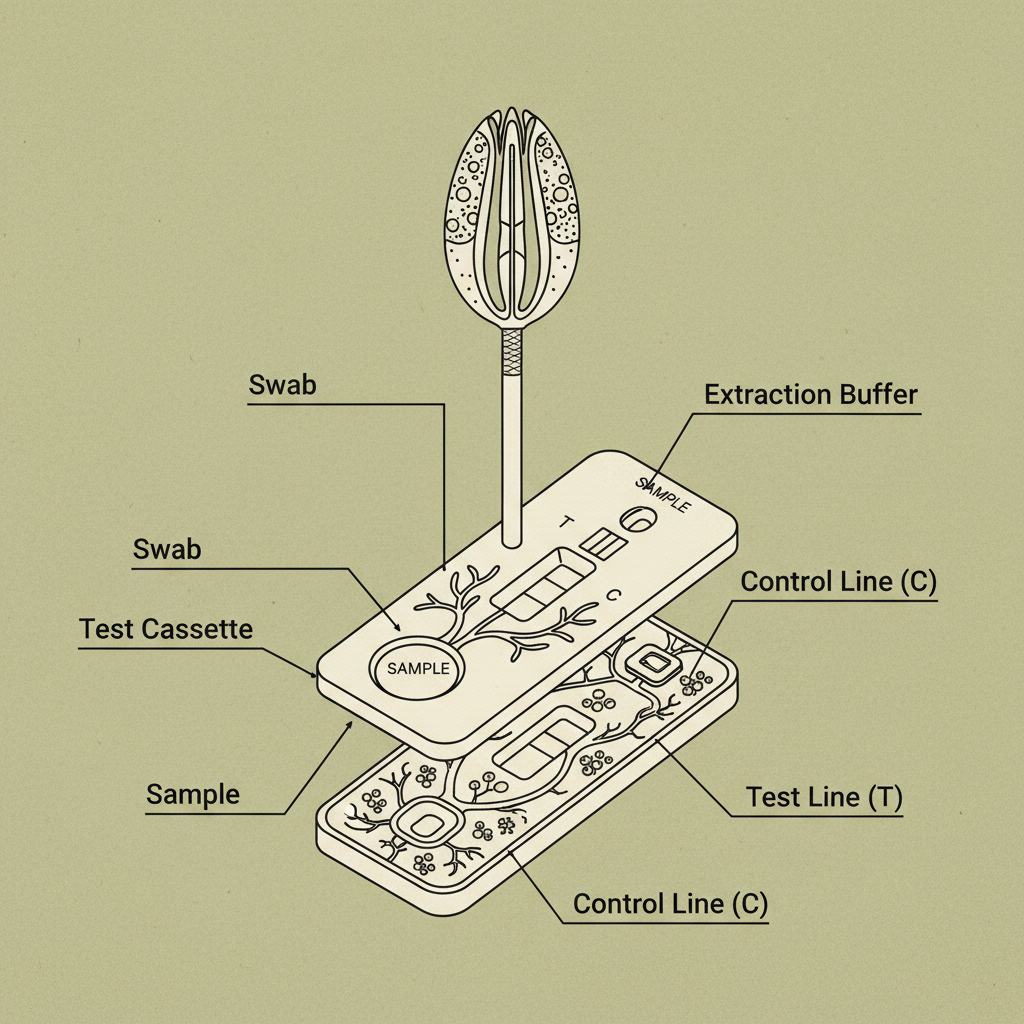
A covid rapid test, also known as a point-of-care test, is a diagnostic tool designed to quickly detect the presence of the SARS-CoV-2 virus, which causes COVID-19. Unlike traditional PCR tests that require laboratory processing, rapid tests typically provide results within 15-30 minutes. These tests generally utilize antigen detection technology, identifying specific viral proteins.
These tests address a crucial need in modern healthcare – accessibility and speed. They're particularly valuable in situations where immediate results are required, such as at the point of care (doctor's offices, clinics), in schools, workplaces, and even for personal use. This accessibility significantly enhances public health surveillance and containment efforts.
The proliferation of covid rapid test technologies reflects a growing demand for decentralized diagnostics, bringing testing capabilities closer to the individuals who need them most. This shift aligns with the broader trend toward personalized medicine and proactive healthcare management.
The core of most covid rapid tests relies on immunochromatographic assays (ICAs). These assays use antibodies to bind to specific viral antigens if present in a sample, typically taken from a nasal or throat swab. The binding of antibodies and antigens creates a visible line on the test strip, indicating a positive result.
Several key components contribute to the effectiveness of these tests, including the specificity of the antibodies used, the quality of the sample collection process, and the sensitivity of the detection system. Factors like temperature and humidity can also impact the accuracy of the results, necessitating careful adherence to manufacturer instructions.
Recent advancements have focused on improving the sensitivity of rapid antigen tests, making them more capable of detecting lower viral loads. This is crucial for identifying asymptomatic or early-stage infections, which are significant drivers of transmission. New technologies are also exploring alternative sample types, such as saliva, to further enhance convenience and accessibility.
The performance of a covid rapid test is influenced by a combination of factors. Sensitivity, the ability to correctly identify positive cases, is paramount, but it’s often lower than that of PCR tests. Specificity, the ability to correctly identify negative cases, is also crucial to minimize false positives.
Other factors include the timing of the test relative to infection – tests are generally most accurate when performed during the peak viral load. Proper sample collection technique is also vital; an inadequate or improperly collected swab can lead to false negatives. Finally, storage and handling of the test kit must adhere to manufacturer guidelines to ensure reliable results.
covid rapid tests are utilized in diverse settings. In healthcare facilities, they enable rapid triage of patients, allowing for swift isolation of infected individuals and minimizing the spread of the virus within hospitals and clinics. They’re also instrumental in screening patients before elective procedures.
Beyond healthcare, these tests are crucial in schools and workplaces for routine screening, helping to identify and isolate cases early, preventing large-scale outbreaks. They are also deployed at mass gatherings, such as concerts and sporting events, to ensure the safety of attendees.
The advantages of covid rapid tests are manifold. Beyond speed and accessibility, they offer significant cost savings compared to PCR testing, particularly when scaled for large-scale screening. The ability to obtain results on-site eliminates the need for complex laboratory infrastructure and reduces turnaround times.
Long-term, these tests contribute to a more resilient public health system, enabling proactive monitoring of infectious diseases and facilitating a rapid response to emerging threats. They empower individuals to take control of their health and make informed decisions, fostering a sense of safety and well-being.
From a social impact perspective, these tests reduce anxiety and uncertainty, allowing for a gradual return to normalcy and minimizing disruptions to daily life. They promote trust in public health initiatives and encourage widespread participation in testing programs.
The future of rapid diagnostics is focused on enhancing sensitivity, reducing cost, and expanding accessibility. Researchers are exploring new technologies, such as CRISPR-based assays and microfluidic devices, to improve the accuracy and speed of detection. Multiplex testing, capable of detecting multiple pathogens simultaneously, is also gaining traction.
Digital integration is another key trend, with the development of smartphone-based testing platforms and data analytics tools to facilitate real-time monitoring of disease outbreaks. These innovations will contribute to a more proactive and data-driven approach to public health management.
Sustainability is also becoming increasingly important, with efforts to develop eco-friendly test kits and reduce waste. Automation and artificial intelligence are poised to streamline the testing process, improving efficiency and reducing human error.
| Challenge | Impact on covid rapid test Performance | Potential Solution | Implementation Difficulty (1-10) |
|---|---|---|---|
| Lower Sensitivity Compared to PCR | Increased False Negative Rate | Development of More Sensitive Antigen Assays | 7 |
| Sample Collection Variability | Inconsistent Test Results | Improved Training and Standardized Protocols | 4 |
| Supply Chain Disruptions | Limited Test Availability | Diversification of Manufacturing and Sourcing | 6 |
| Data Interpretation Challenges | Misleading Results | Development of User-Friendly Data Analytics Tools | 5 |
| Limited Accessibility in Remote Areas | Unequal Access to Testing | Deployment of Mobile Testing Units | 8 |
| Cost Considerations | Barriers to Widespread Testing | Government Subsidies and Affordable Test Options | 6 |
While PCR tests remain the gold standard for accuracy, covid rapid tests offer a good balance of speed and sensitivity. Generally, rapid tests are less sensitive than PCR, meaning they might miss some cases, particularly those with low viral loads. However, they excel at identifying individuals who are most infectious, making them useful for quick screening and containment.
A negative result on a covid rapid test does not completely rule out infection, especially if you're experiencing symptoms. It's recommended to follow up with a PCR test to confirm the result. Continue to practice preventative measures like masking and social distancing while awaiting your PCR results and consult a healthcare professional if your symptoms worsen.
The validity of a covid rapid test result depends on the specific requirements of the situation. Generally, results are considered valid for a short period, typically 24-48 hours, especially for travel or event entry. It's crucial to check the specific guidelines of the organization or destination requiring the test.
Yes, you can use a covid rapid test even if you’re asymptomatic. While symptomatic individuals are more likely to test positive, asymptomatic individuals can still carry and transmit the virus. Regular testing, even without symptoms, can help identify and prevent further spread.
Proper storage of a covid rapid test kit is essential for ensuring accurate results. Always store the kit according to the manufacturer's instructions, typically in a cool, dry place away from direct sunlight and extreme temperatures. Check the expiration date before use.
Accurate sample collection is vital. Carefully follow the instructions provided with the covid rapid test kit. Typically, this involves gently swirling a swab inside your nostrils or throat for a specified duration and depth. Avoid eating, drinking, or touching your nose for at least 30 minutes before sample collection.
The covid rapid test has fundamentally changed our approach to infectious disease management. Its speed, accessibility, and affordability have made it an indispensable tool for controlling outbreaks, protecting vulnerable populations, and facilitating a safe return to normalcy. The ability to quickly identify and isolate cases has proven crucial in mitigating the spread of the virus and easing the burden on healthcare systems.
Looking ahead, continued innovation in rapid diagnostic technologies, coupled with robust public health infrastructure, will be essential for preparing for future pandemics. Increased investment in research and development, alongside equitable access to testing resources, will strengthen our collective ability to respond effectively to emerging health threats. Visit our website at www.prisesbio.com to learn more about our covid rapid test solutions.