
- Afrikaans
- Albanian
- Amharic
- Arabic
- Armenian
- Azerbaijani
- Basque
- Belarusian
- Bengali
- Bosnian
- Bulgarian
- Catalan
- Cebuano
- China
- China (Taiwan)
- Corsican
- Croatian
- Czech
- Danish
- Dutch
- English
- Esperanto
- Estonian
- Finnish
- French
- Frisian
- Galician
- Georgian
- German
- Greek
- Gujarati
- Haitian Creole
- hausa
- hawaiian
- Hebrew
- Hindi
- Miao
- Hungarian
- Icelandic
- igbo
- Indonesian
- irish
- Italian
- Japanese
- Javanese
- Kannada
- kazakh
- Khmer
- Rwandese
- Korean
- Kurdish
- Kyrgyz
- Lao
- Latin
- Latvian
- Lithuanian
- Luxembourgish
- Macedonian
- Malgashi
- Malay
- Malayalam
- Maltese
- Maori
- Marathi
- Mongolian
- Myanmar
- Nepali
- Norwegian
- Norwegian
- Occitan
- Pashto
- Persian
- Polish
- Portuguese
- Punjabi
- Romanian
- Russian
- Samoan
- Scottish Gaelic
- Serbian
- Sesotho
- Shona
- Sindhi
- Sinhala
- Slovak
- Slovenian
- Somali
- Spanish
- Sundanese
- Swahili
- Swedish
- Tagalog
- Tajik
- Tamil
- Tatar
- Telugu
- Thai
- Turkish
- Turkmen
- Ukrainian
- Urdu
- Uighur
- Uzbek
- Vietnamese
- Welsh
- Bantu
- Yiddish
- Yoruba
- Zulu
PRISES Biotechnology is een op R&D gebaseerde fabrikant, die zich bezighoudt met de ontwikkeling, productie en handel in in vitro diagnostische reagentia (IVD) en medische apparatuur, die is goedgekeurd voor de productie en handel in IVD-producten van NMPA (CFDA) en werkt onder het kwaliteitssysteem van ISO 13485, de meeste van de producten zijn gecertificeerd met CE-markering.
belangrijk
producten
-

Uncut sheet
Uncut sheet
Ongesneden vel voor snelle test
-
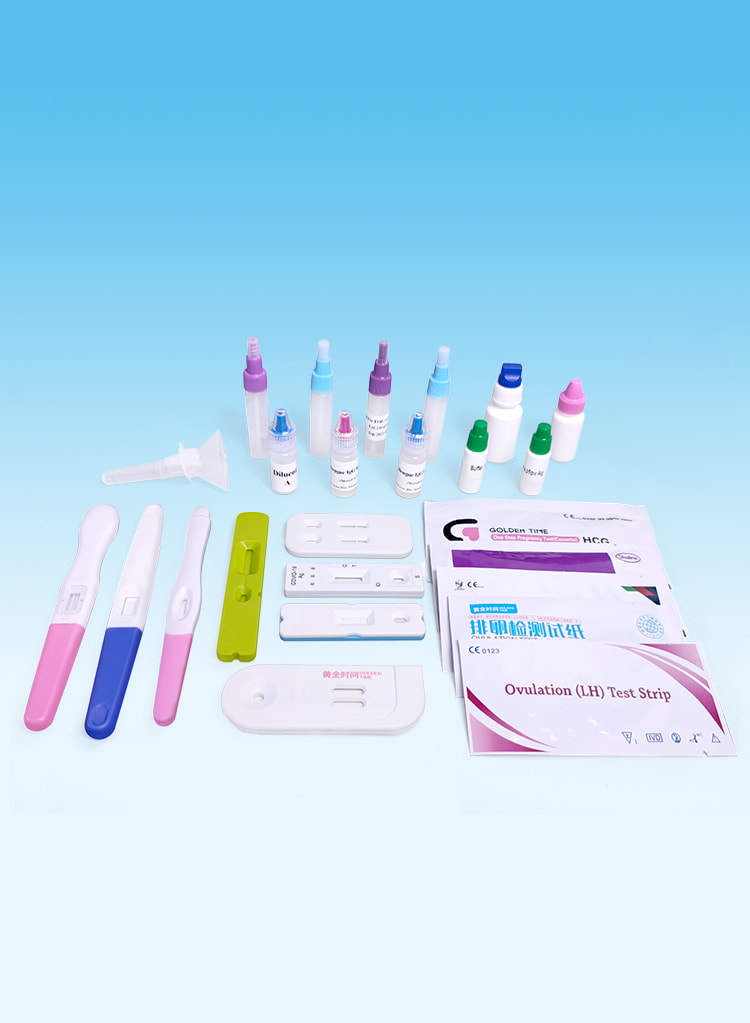
Plastic Cassette
Plastic Cassette
Kunststof cassette voor snelle test
-

Testkit voor infectieziekten
Testkit voor infectieziekten
Detects STDs, Hepatitis, H. pylori, Malaria, Dengue, Typhoid, Influenza and other diseases.
-

Vruchtbaarheidstesten
Vruchtbaarheidstesten
Effective detection of pregnancy and ovulation.
over
PRISES
Our factory is founded in 2012 and located in Gaobeidian City, which is near Xiongan New Area and Beijing. It covers an area of 3,000 square meters, including class 1000,000 clean workshop with 1000 square meters, class 10 thousands of microbiological testing room with 500 square meters, well-equipped quality inspection rooms, research and development laboratories, etc.
-

10+
Business growth
10+
Business growthEstablished 11 years ago, the company has become a well-established and dynamic business through continuous efforts and improvements.
Details bekijken -

100000
Clean workshop
100000
Clean workshopClass 100,000 clean workshop 1000 square metres, Class 10,000 microbiological test room 500 square metres, fully equipped quality control room, R&D laboratory, laboratory, etc.
Details bekijken -

30+
Research staff
30+
Research staffThe company has a research and development team of 35 people who drive the technology and support the development of new products for the company.
Details bekijken -

20+
Consultant
20+
ConsultantWe provide the highest level of service to our customers, whether it's answering questions, solving problems or providing valuable advice, we deal with it promptly.
Details bekijken -

100+
Professionals
100+
ProfessionalsThe company has a large team of professionals responsible for managing, coordinating and controlling the operations of the various functions.
Details bekijken
nieuws en informatie
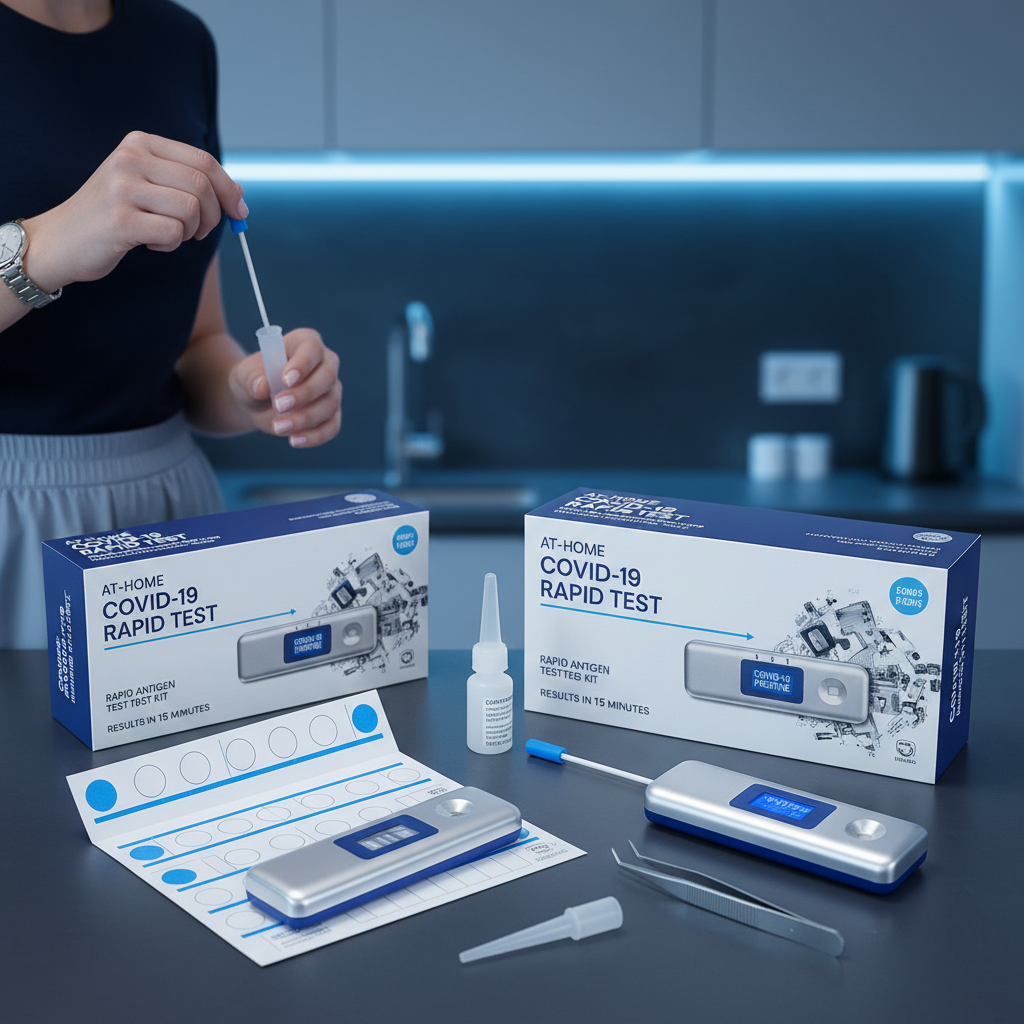
Buy at home covid test rapid covid test kit antigen test covid19
In an era where health vigilance is more important than ever, having a reliable at home covid test has become a household necessity. Whether you are preparing for travel, returning to work, or simply feeling under the weather, the ability to screen yourself quickly and privately is invaluable. Modern diagnostic technology has evolved to provide professional-grade accuracy within the comfort of your living room. This guide will explore the nuances of rapid testing, helping you understand how to select the most effective kits to keep your family and community safe.

Covid19 Antigent Test Kit Cassette Covid Test Rapid Diagnostic Covid 19 Antigen Rapid Test
In the evolving landscape of global health, having access to reliable and fast screening tools is essential for safety and peace of mind. The covid-19 antigent test kit cassette has become a cornerstone of pandemic management, providing a decentralized way to monitor viral presence. These diagnostic tools are designed for ease of use, allowing individuals and healthcare providers to detect the SARS-CoV-2 virus without the immediate need for a laboratory. By utilizing highly specific antibodies, these kits offer a rapid response that is critical for timely isolation and treatment. In this guide, we will explore the technical advantages and practical applications of modern rapid testing solutions.

Comprehensive Guide to Antigen Covid Test Covid Saliva Test and Covid Test Kit 19 Options
In the ongoing effort to manage and mitigate the spread of COVID-19, rapid and reliable testing is crucial. Different types of COVID-19 tests are available, each with its own advantages and use cases. This article will explore the key differences between antigen COVID tests, COVID saliva tests, and COVID test kits, providing a comprehensive guide to help you understand which option is best for your needs.





