The global need for rapid and accurate disease detection has never been greater, and the covid-19 rapid test kit plays a critical role in meeting this demand. These kits offer a fast, convenient, and often cost-effective method for identifying infections, enabling prompt medical intervention and helping to control outbreaks. Understanding their functionality, limitations, and proper use is paramount for healthcare professionals, public health organizations, and individuals alike.
The rise of infectious diseases, compounded by global travel and population density, necessitates widespread testing capabilities. Traditional laboratory-based testing can be time-consuming, requiring specialized equipment and trained personnel. covid-19 rapid test kit offer a solution by providing results within minutes, facilitating quicker decision-making regarding isolation, treatment, and contact tracing. This speed is invaluable in mitigating the spread of illness.
Beyond immediate health benefits, the widespread availability of these kits has broader societal impacts. They support economic stability by allowing businesses and schools to remain open safely, and they empower individuals to take proactive steps to protect themselves and their communities. Effective management of public health crises hinges on accessible and reliable testing solutions like the covid-19 rapid test kit.
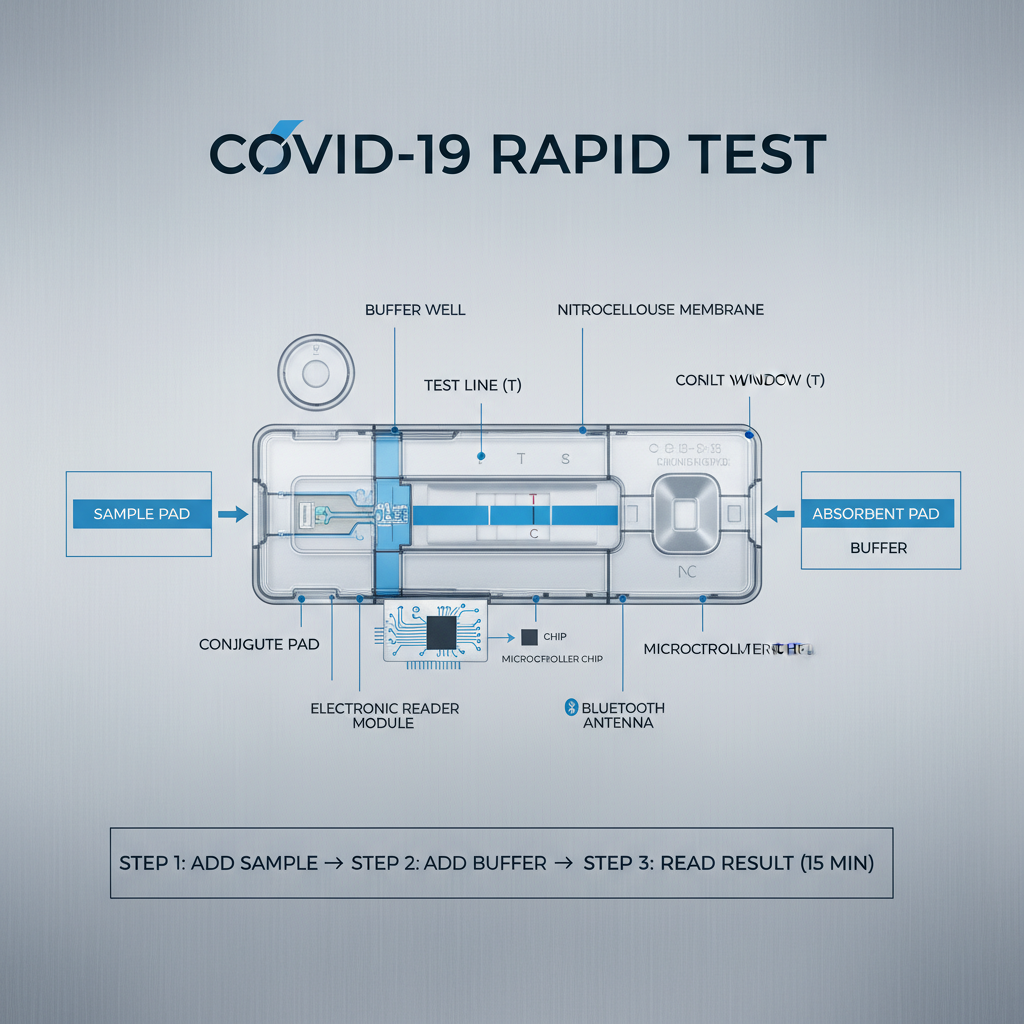
covid-19 rapid test kit utilize different methodologies, primarily focusing on antigen or antibody detection. Antigen tests identify specific proteins from the virus itself, while antibody tests detect the body’s immune response to the virus. The fundamental principle relies on biochemical reactions that produce a visible signal indicating the presence or absence of the target substance.
Understanding the underlying science behind these tests is crucial for interpreting results accurately. Factors like sensitivity (ability to detect true positives) and specificity (ability to detect true negatives) directly impact the reliability of the kits. Variations in test design and manufacturing processes can also influence performance characteristics, highlighting the importance of quality control and regulatory oversight.
The COVID-19 pandemic dramatically increased the global demand for rapid diagnostic testing, driving significant growth in the covid-19 rapid test kit market. According to the World Health Organization (WHO), widespread testing is a cornerstone of pandemic response, alongside vaccination and public health measures. This surge in demand necessitated rapid innovation and scaling of production capabilities.
The market is characterized by a diverse range of manufacturers, from established multinational corporations to emerging biotechnology companies. Regions with high population density and limited access to traditional laboratory infrastructure have particularly benefited from the availability of these kits. The United Nations Sustainable Development Goals (SDGs), specifically those related to health and well-being, are directly impacted by the effectiveness of rapid diagnostic testing programs.
The increasing demand isn't limited to pandemic responses; rapid testing is also becoming integral to routine healthcare, workplace safety protocols, and event management. This broadening application base ensures the continued relevance and growth of the covid-19 rapid test kit market.
A covid-19 rapid test kit is a diagnostic tool designed to detect the presence of SARS-CoV-2, the virus that causes COVID-19, within a short timeframe—typically 15-30 minutes. Unlike traditional PCR tests which require sophisticated laboratory equipment and trained personnel, rapid tests can often be administered and interpreted on-site, making them ideal for point-of-care settings.
These kits operate on principles of immunoassay technology. Most commonly, they utilize lateral flow technology, similar to a home pregnancy test. A sample (typically a nasal swab) is introduced to a test strip containing antibodies that bind to specific viral antigens or antibodies produced by the body. The presence of a colored line indicates a positive result.
The widespread adoption of covid-19 rapid test kit represents a significant advancement in public health, enabling quicker diagnoses and facilitating faster responses to outbreaks. They are instrumental in both individual healthcare decisions and broader public health management strategies.
Key components of a covid-19 rapid test kit include the test strip, sample collection swab, extraction buffer, and a disposable dropper or pipette. The test strip is the core of the device, containing antibodies and a detection system that generates a visible result. The quality of these components significantly impacts the test's accuracy and reliability.
Performance characteristics, such as sensitivity and specificity, are critical metrics. Sensitivity refers to the test’s ability to correctly identify positive cases, while specificity measures its ability to correctly identify negative cases. A high sensitivity minimizes false negatives, while high specificity minimizes false positives. Understanding these parameters is essential for appropriate interpretation of test results.
covid-19 rapid test kit are deployed across a wide spectrum of settings, including healthcare facilities, schools, workplaces, travel hubs, and community testing sites. Their portability and ease of use make them particularly valuable in remote areas with limited access to traditional laboratory services.
In post-disaster relief operations, rapid testing is crucial for identifying and isolating infected individuals, preventing further spread of the virus within vulnerable populations. Similarly, in remote industrial zones, covid-19 rapid test kit help maintain workforce health and operational continuity. They also play a pivotal role in pre-travel screening, reducing the risk of importing new cases.
The primary advantage of covid-19 rapid test kit lies in their speed and convenience. Results are available within minutes, enabling prompt medical decisions and reducing the time required for isolation and treatment. This rapid turnaround time is crucial for controlling outbreaks and minimizing the spread of infection.
Beyond immediate health benefits, widespread access to rapid testing promotes a sense of safety and empowerment, allowing individuals to proactively manage their health and protect their communities. The development and deployment of these kits have also spurred innovation in diagnostic technology, paving the way for more efficient and accessible healthcare solutions in the future. The long-term impact is increased preparedness for future pandemics and improved public health infrastructure.
Furthermore, the cost-effectiveness of rapid testing, compared to more elaborate PCR tests, makes it a sustainable solution for routine monitoring and surveillance. This affordability is particularly important in resource-limited settings.
Future advancements in covid-19 rapid test kit technology are focused on improving sensitivity, specificity, and multiplexing capabilities—the ability to detect multiple pathogens simultaneously. Nanotechnology and microfluidics are being explored to enhance detection limits and reduce testing time. Digital integration, including smartphone-based readers and data analytics platforms, is also gaining traction.
The development of self-administered tests, coupled with telehealth platforms, promises to further democratize access to rapid diagnostics. Artificial intelligence (AI) and machine learning (ML) algorithms are being employed to analyze test results and identify emerging variants. These innovations will play a crucial role in strengthening global pandemic preparedness and improving public health outcomes.
Sustainability is also becoming a key consideration, with manufacturers exploring eco-friendly materials and reducing waste associated with kit disposal. Emphasis on ethical data handling and privacy protection will be critical as digital integration expands.
| Technology Type | Sensitivity Range | Specificity Range | Turnaround Time (minutes) |
|---|---|---|---|
| Lateral Flow Assay | 60-80% | 95-99% | 15-30 |
| Isothermal Amplification | 85-95% | 98-100% | 30-60 |
| Molecular Point-of-Care | 90-98% | 99-100% | 45-90 |
| Electrochemical Biosensors | 75-90% | 97-99% | 20-40 |
| Microfluidic Platforms | 80-95% | 98-99% | 25-50 |
| Digital Immunoassay | 70-85% | 95-98% | 10-20 |
While PCR tests are generally considered the "gold standard" for accuracy, covid-19 rapid test kit offer a good balance between speed and reliability. Rapid antigen tests typically have a sensitivity of 60-80% and a specificity of 95-99%, meaning they may miss some positive cases (false negatives) but are very good at correctly identifying negative cases (true negatives). PCR tests are more sensitive but take longer to process. The best test depends on the specific situation and testing goals.
Proper storage is critical for maintaining the accuracy of the test. Generally, kits should be stored at room temperature, away from direct sunlight and extreme temperatures. It's important to check the expiration date on the kit before use. Avoid storing kits in humid environments, as moisture can damage the components. Always follow the specific storage instructions provided by the manufacturer.
If you test positive, it’s crucial to isolate yourself immediately to prevent further spread of the virus. Follow the current guidelines from your local health authorities regarding isolation and quarantine protocols. Contact your healthcare provider for guidance on next steps, including potential treatment options. It’s also advisable to inform your close contacts so they can get tested and take appropriate precautions.
No, absolutely not. All components of a covid-19 rapid test kit are designed for single-use only. Reusing any component can lead to inaccurate results and potentially spread contamination. Ensure you use a fresh swab for each test and properly dispose of all used materials according to local regulations. Using a new and sterile device is key for maintaining result validity.
Antigen tests detect the presence of viral proteins (antigens) in the body, indicating a current infection. They are most accurate when the viral load is high, typically during the symptomatic phase. Antibody tests, on the other hand, detect antibodies produced by the immune system in response to the virus. These tests can indicate past infection, but they may not be useful for diagnosing a current infection.
Many covid-19 rapid test kit are approved for use in children, but it’s important to check the manufacturer's instructions and age recommendations. Sample collection methods may vary for children, often requiring assistance from an adult. It's essential to ensure the child is comfortable and cooperative during the testing process. If you have concerns, consult with a pediatrician.
In conclusion, covid-19 rapid test kit have revolutionized disease detection, offering a fast, convenient, and accessible solution for identifying infections. Their widespread adoption has had a profound impact on public health, enabling quicker responses to outbreaks, supporting economic stability, and empowering individuals to protect themselves and their communities. Continuous innovation in this field promises even more accurate, efficient, and user-friendly testing options in the future.
Looking ahead, investment in research and development, coupled with robust regulatory oversight, is crucial for ensuring the quality and reliability of these kits. Expanding access to testing, particularly in resource-limited settings, remains a priority. By embracing innovation and prioritizing public health, we can leverage the power of covid-19 rapid test kit to build a more resilient and prepared global community. Visit our website: www.prisesbio.com
