The global demand for rapid and reliable diagnostic tools is constantly growing, with pregnancy tests representing a significant portion of this market. The accuracy and accessibility of these tests are crucial for early detection and informed healthcare decisions. Understanding the intricacies of pregnancy test strip urine – its manufacturing, quality control, and application – is paramount for manufacturers, healthcare providers, and ultimately, expectant mothers worldwide.
In the medical device industry, particularly within in-vitro diagnostics, the consistent performance and sensitivity of pregnancy tests are non-negotiable. The pregnancy test strip urine market is driven by factors like increasing awareness of reproductive health, rising disposable incomes, and advancements in test technology. Efficient production and distribution chains are vital to meet the global needs.
The development and refinement of pregnancy test strip urine have had a profound impact on early prenatal care. These tests empower individuals to take proactive steps regarding their health and reproductive planning. This contributes to better pregnancy outcomes and overall public health.


The fundamental principle behind pregnancy test strip urine relies on detecting the presence of human chorionic gonadotropin (hCG) in urine. hCG is a hormone produced during pregnancy, making its detection a reliable indicator. These tests utilize antibodies that specifically bind to hCG, creating a visible signal.
This binding reaction is often visualized through a color change on the test strip, indicating a positive result. The sensitivity of the test, determined by the concentration of hCG it can detect, directly impacts its accuracy, particularly in the early stages of pregnancy.
The manufacturing of pregnancy test strip urine involves a complex series of steps, beginning with the production of antibodies specific to hCG. These antibodies are then immobilized onto a porous membrane, typically made of nitrocellulose. Careful control of the antibody concentration and distribution is vital for test performance.
Rigorous quality control measures are implemented throughout the process, including testing for sensitivity, specificity, and stability. Batch-to-batch consistency is ensured through standardized protocols and meticulous documentation. This guarantees reliable results for end-users.
Adherence to international standards, such as ISO 13485, is crucial for manufacturers aiming to distribute their products globally. These standards ensure the quality management system is robust and consistently delivers safe and effective pregnancy tests.
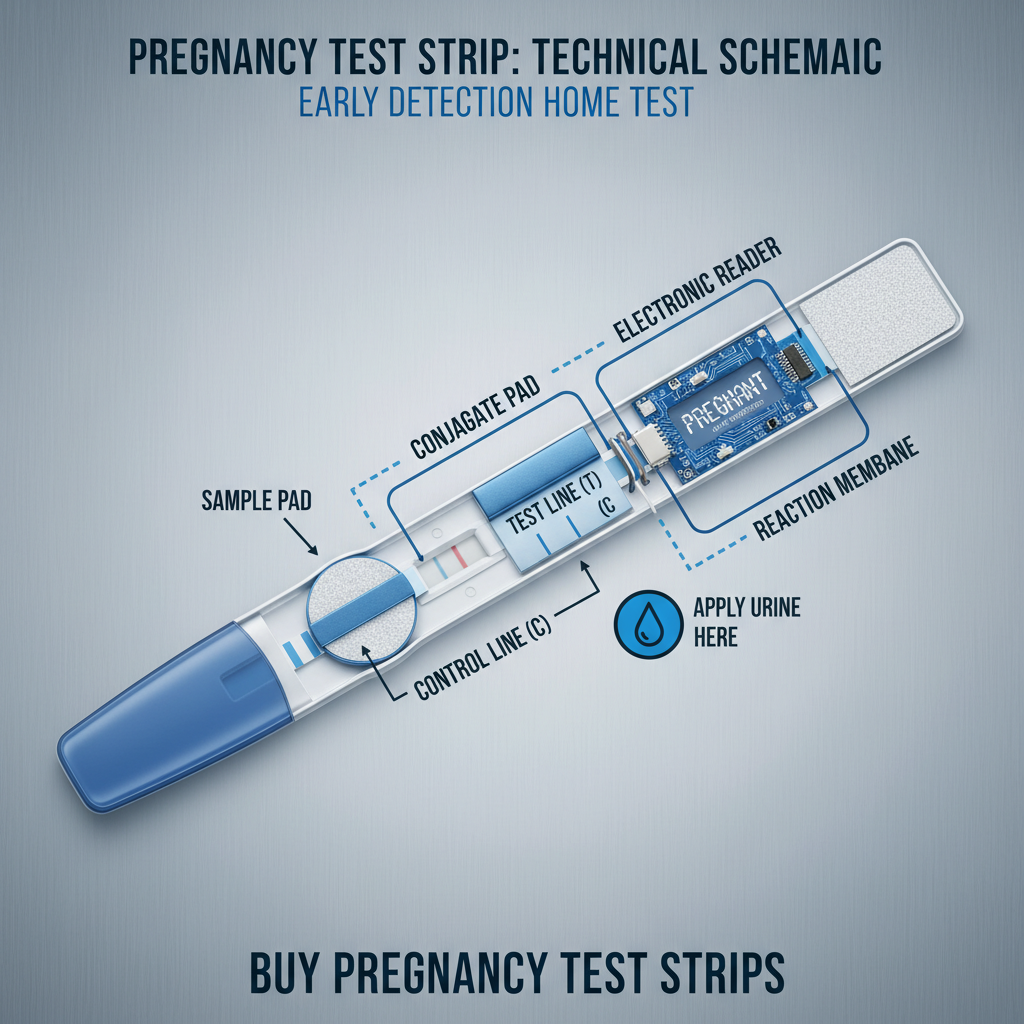
A typical pregnancy test strip urine comprises several essential components. These include the sample pad, which draws urine into the test; the conjugate pad, containing the hCG antibodies; the test line, where the antibody-hCG complex binds to create a visible signal; and the control line, confirming the test has functioned correctly.
The backing card provides structural support for the strip, while the absorbent pad ensures unidirectional flow of the urine sample. The quality and interaction of each component are critical for achieving accurate and reliable test results.
Variations in these components, such as the type of antibody used or the membrane material, can impact the test's sensitivity and specificity. Continuous research and development efforts are focused on optimizing these components for improved performance.
The distribution of pregnancy test strip urine is a global endeavor, involving complex logistics and regulatory compliance. Manufacturers must navigate varying requirements across different countries, including registration processes, labeling standards, and quality control certifications.
Developed markets like the United States and Europe have stringent regulations enforced by agencies like the FDA and CE marking authorities. Emerging markets often have less-developed regulatory frameworks, presenting both challenges and opportunities for manufacturers.
pregnancy test strip urine find widespread application in both clinical and home settings. In clinical laboratories, they are used for confirming pregnancy during routine check-ups or as part of fertility investigations.
The convenience and affordability of home pregnancy tests have made them the first line of detection for many individuals. These tests empower women to take control of their reproductive health and seek early prenatal care if needed.
The primary advantages of pregnancy test strip urine are their ease of use, rapid results, and relatively low cost. They provide a quick and convenient method for confirming pregnancy, particularly in situations where immediate clinical access is limited.
However, these tests also have limitations. False negatives can occur if the test is taken too early in pregnancy, before hCG levels are sufficiently high. False positives, while rare, can result from certain medical conditions or medications.
It's important to note that these tests are intended as a screening tool and should be confirmed by a healthcare professional for definitive diagnosis and appropriate prenatal care.
Future trends in pregnancy test strip urine development focus on enhancing sensitivity, reducing the likelihood of false results, and incorporating digital connectivity. Researchers are exploring new antibody technologies and signal amplification methods to improve test accuracy.
The integration of smartphone apps and Bluetooth connectivity is enabling the creation of digital pregnancy tests that provide personalized guidance and track test results over time. This opens up possibilities for remote monitoring and data analysis.
Furthermore, there is growing interest in developing multiplex tests that can simultaneously detect pregnancy and other biomarkers, offering a more comprehensive assessment of maternal health.
| Sensitivity (mIU/mL) | Specificity (%) | Read Time (minutes) | Cost per Unit ($) |
|---|---|---|---|
| 20 mIU/mL | 99.5% | 5 | 1.50 |
| 25 mIU/mL | 99.2% | 3 | 1.25 |
| 10 mIU/mL | 99.8% | 2 | 2.00 |
| 30 mIU/mL | 99.0% | 5 | 1.00 |
| 15 mIU/mL | 99.6% | 4 | 1.75 |
| 22 mIU/mL | 99.3% | 3 | 1.30 |
Home pregnancy tests are generally highly accurate, around 99% when used correctly. Accuracy depends on following instructions carefully, testing at the right time (after a missed period), and the sensitivity of the test. Factors like diluted urine or testing too early can lead to false negatives, while rare cases of medication interference can cause false positives. If results are unclear or unexpected, consult a healthcare professional.
The most accurate results are obtained by taking a pregnancy test at least one day after a missed period. This allows sufficient time for hCG levels to rise to detectable levels. Some highly sensitive tests can detect pregnancy a few days before a missed period, but the accuracy is lower at that stage. Following the test instructions is crucial.
Certain medications, particularly those containing hCG (like fertility drugs), can lead to a false positive result. Rarely, certain medical conditions like ovarian cysts or ectopic pregnancies can also affect test outcomes. Always inform your healthcare provider about any medications you are taking or medical conditions you have when interpreting a pregnancy test result.
A faint positive line indicates a low level of hCG, which could mean you are very early in your pregnancy. It is recommended to retest in a few days, using first-morning urine which has a higher concentration of hCG, to confirm the result. If the line becomes darker, it likely confirms pregnancy; if it remains faint or disappears, consult a doctor.
If you receive a negative result but still suspect pregnancy, retest in a few days, preferably using first-morning urine. If the test remains negative, consult a healthcare provider for further evaluation. They can perform a blood test, which is more sensitive and can detect pregnancy earlier than urine tests.
Pregnancy test strips should be stored in a cool, dry place, away from direct sunlight and moisture. It is important to check the expiration date on the packaging before using the test. Expired tests may not provide accurate results. Keep the sealed pouch closed until ready to use.
In conclusion, pregnancy test strip urine represents a critical diagnostic tool with far-reaching implications for women's health and reproductive planning. Their affordability, accessibility, and ease of use have made them a cornerstone of early prenatal care worldwide. Understanding the underlying principles, manufacturing processes, and potential limitations of these tests is crucial for ensuring accurate results and informed healthcare decisions.
Looking ahead, continued innovation in this field promises even more sensitive, reliable, and digitally integrated pregnancy tests. Focusing on improving test accuracy, expanding accessibility to underserved populations, and exploring multiplex testing capabilities will further enhance the impact of pregnancy test strip urine on global health outcomes.