The global landscape of infectious disease monitoring relies heavily on accurate and timely diagnostic testing. Understanding the nuances of test covid-19 igg igm is crucial for public health initiatives, clinical decision-making, and tracking the progression of immunity within populations. This knowledge informs effective strategies for containment, treatment, and vaccination programs, ultimately safeguarding communities worldwide.
The increasing demand for rapid and reliable diagnostic solutions, especially in the wake of global health crises like the COVID-19 pandemic, has spotlighted the importance of serological testing. Test covid-19 igg igm specifically aids in determining past infection by identifying antibodies developed in response to the virus. This information is vital for assessing population-level immunity and understanding the long-term impact of the disease.
Furthermore, the availability of affordable and accessible test covid-19 igg igm empowers individuals to take proactive control of their health. It allows for informed decisions about personal safety, return-to-work protocols, and participation in social activities, contributing to a more resilient and informed society. This testing plays a key role in navigating the ongoing challenges posed by evolving viral strains and potential future outbreaks.


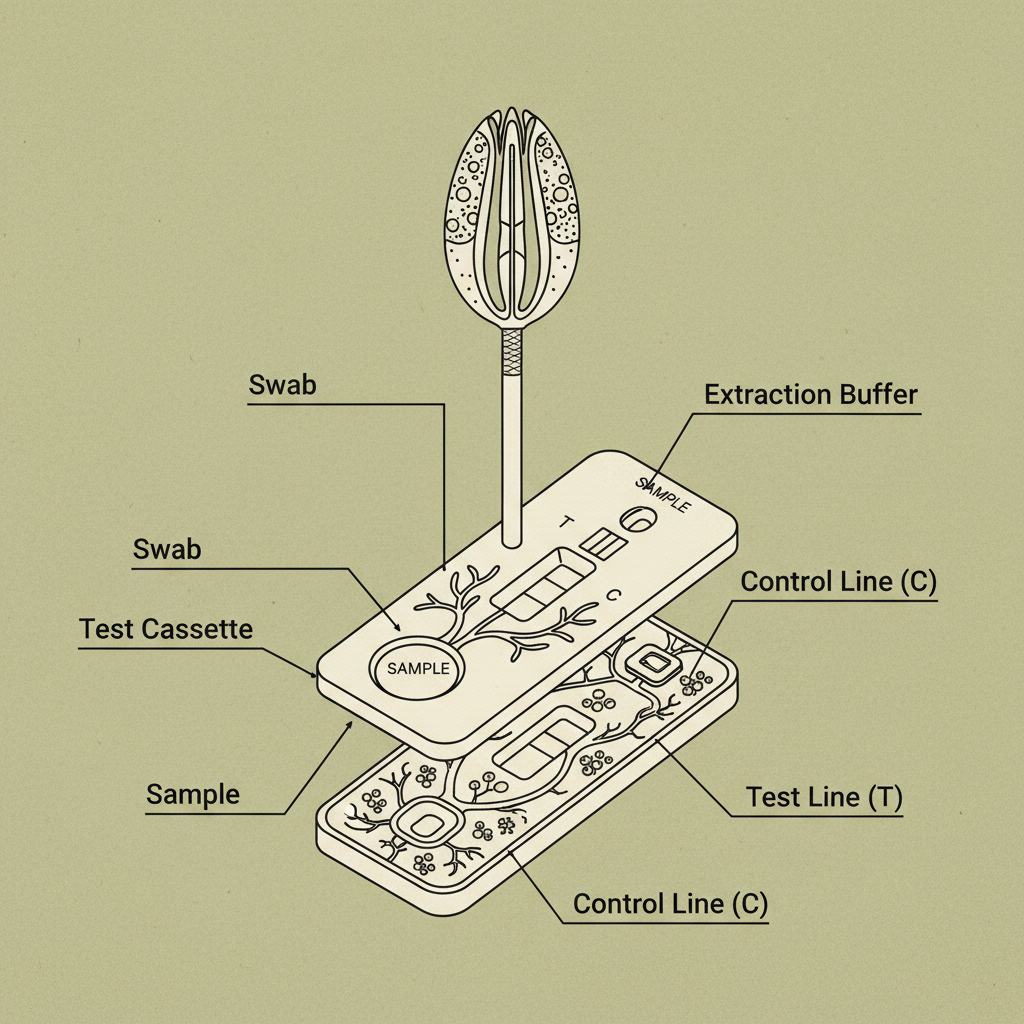
At its core, test covid-19 igg igm involves detecting the presence of specific antibodies in a blood sample. IgG (Immunoglobulin G) antibodies typically appear later in the infection process and indicate past exposure, providing insight into long-term immunity. IgM (Immunoglobulin M) antibodies are usually the first to appear, suggesting a recent or active infection. The combined analysis of both IgG and IgM levels is instrumental in accurately determining the stage and history of a COVID-19 infection.
The development of reliable test covid-19 igg igm assays has been a collaborative effort involving researchers, diagnostic manufacturers, and regulatory bodies. Different testing methodologies, such as enzyme-linked immunosorbent assays (ELISAs) and lateral flow immunoassays, offer varying levels of sensitivity and specificity, impacting the overall accuracy of results.
The COVID-19 pandemic, declared by the World Health Organization (WHO) in March 2020, has underscored the critical need for widespread diagnostic testing. As of late 2023, over 700 million confirmed cases of COVID-19 have been reported globally, with millions of lives lost. This has placed immense pressure on healthcare systems worldwide. Test covid-19 igg igm data plays a vital role in tracking the pandemic’s evolution, identifying hotspots, and assessing the effectiveness of public health interventions.
Data from the United Nations and various national health agencies indicate a continued need for serological surveillance even as acute infection rates fluctuate. Understanding the prevalence of antibodies helps inform vaccination strategies and identify populations at higher risk of severe disease. The World Bank has also highlighted the economic impact of the pandemic, emphasizing the importance of rapid diagnostics for enabling a safe and sustainable return to economic activity. The need for accurate test covid-19 igg igm remains paramount.
The ISO (International Organization for Standardization) has established standards for diagnostic testing, including those used for test covid-19 igg igm, to ensure quality and reliability across different manufacturers and regions. Compliance with these standards is crucial for building trust in test results and supporting informed public health decision-making. The challenge lies in ensuring equitable access to these standardized tests, particularly in low- and middle-income countries.
Simply put, test covid-19 igg igm is a blood test used to detect antibodies produced by the body in response to a SARS-CoV-2 (the virus that causes COVID-19) infection. IgM antibodies usually appear within a week of initial infection, while IgG antibodies develop later and can persist for months or even years, indicating longer-term immunity. The test doesn't detect the virus itself, but rather the immune system’s response to it.
This type of testing is directly relevant to modern public health initiatives focused on disease surveillance and immunity assessment. It differs from PCR tests, which detect the presence of the virus's genetic material. PCR tests are used to diagnose active infections, while test covid-19 igg igm tests help determine past infections or the level of immunity.
In the humanitarian context, test covid-19 igg igm can be valuable for identifying individuals who may have prior immunity within vulnerable populations, helping to prioritize vaccination efforts and resource allocation. Furthermore, it is being used in research to understand the duration of immunity after infection or vaccination, which informs strategies for booster shots and long-term protection.
Several factors can influence the accuracy of test covid-19 igg igm results. One crucial element is the timing of the test relative to infection. Testing too early, before antibodies have developed, can lead to false negatives. Conversely, testing too late might result in diminishing antibody levels that are difficult to detect. Individual immune responses also vary; some people may produce fewer antibodies than others.
The quality and sensitivity of the test itself are also paramount. Different manufacturers employ different methodologies and antibody targets, resulting in varying levels of accuracy. It is essential to utilize tests that have been validated by reputable organizations and demonstrate high sensitivity and specificity.
In public health, test covid-19 igg igm is widely used for seroprevalence studies, which determine the proportion of a population that has antibodies against the virus. This data informs vaccination strategies, resource allocation, and the monitoring of herd immunity. These studies are vital for guiding public health policies and assessing the effectiveness of interventions.
Clinically, these tests aid in diagnosing past infections, assessing the severity of illness, and understanding the immune response in individual patients. In research, test covid-19 igg igm is employed to study the long-term effects of COVID-19, the duration of immunity, and the effectiveness of different vaccines. In post-disaster relief operations, rapid antibody testing can help identify individuals with prior immunity, allowing for more targeted resource allocation.
The advantages of test covid-19 igg igm are numerous. They offer a relatively cost-effective way to assess population-level immunity compared to repeated PCR testing. They provide insights into the duration of protection following infection or vaccination, which is crucial for guiding booster shot schedules. Furthermore, the results can empower individuals to make informed decisions about their health and safety. This is particularly important for vulnerable populations.
The long-term value lies in building a more resilient public health infrastructure. By understanding the dynamics of antibody responses, we can better prepare for future outbreaks and pandemics. The data generated from these tests contributes to a growing body of knowledge that informs vaccine development, treatment strategies, and public health policies. A proactive approach based on serological surveillance enhances preparedness and minimizes the impact of emerging infectious diseases.
Future trends point toward the development of more sophisticated and accurate test covid-19 igg igm assays, including multiplex assays capable of detecting antibodies against multiple variants simultaneously. Point-of-care testing, allowing for rapid results in decentralized settings, is also gaining traction. Automation and digital technologies are streamlining the testing process, improving efficiency and reducing the risk of errors.
The integration of artificial intelligence (AI) and machine learning (ML) is poised to enhance the interpretation of results, identifying subtle patterns and predicting potential outbreaks. Furthermore, research into novel antibody detection methods, such as microfluidic devices and biosensors, promises even faster and more sensitive testing capabilities. These innovations will contribute to a more proactive and data-driven approach to pandemic preparedness.
Sustainability is also becoming a key focus, with efforts to develop eco-friendly testing kits and reduce waste generation. The move towards digital health solutions will facilitate data sharing and collaboration, enabling real-time monitoring of antibody prevalence and rapid response to emerging threats.
| Challenge | Impact on test covid-19 igg igm Accuracy | Potential Solutions | Implementation Difficulty (1-10) |
|---|---|---|---|
| Cross-Reactivity with Other Antibodies | False positives due to antibodies from prior infections. | Develop highly specific assays targeting unique SARS-CoV-2 epitopes. | 7 |
| Variations in Antibody Response | Inconsistent results across individuals. | Standardize testing protocols and establish reference ranges based on diverse populations. | 6 |
| Limited Access in Resource-Constrained Settings | Unequal access to testing and surveillance data. | Develop affordable and portable testing solutions. | 8 |
| Emergence of New Variants | Potential for reduced antibody detection due to mutations. | Regularly update assays to target conserved epitopes of new variants. | 5 |
| Sample Collection and Storage | Degradation of antibodies during transport and storage. | Implement standardized protocols for sample collection, handling, and storage. | 4 |
| Data Interpretation and Standardization | Inconsistent reporting and interpretation of results. | Develop standardized reporting guidelines and training programs. | 6 |
IgM antibodies appear early in an infection, indicating a recent or active infection. IgG antibodies develop later and can stay in the body for months, suggesting past exposure and potential immunity. Test covid-19 igg igm combines these two measurements to help determine the stage and history of an infection. The presence of only IgG suggests a past infection, while IgM indicates a recent one. Combined positivity suggests an active or recent infection.
Accuracy varies depending on the specific test, the timing of testing, and individual factors. High-quality assays, when performed correctly, generally have high sensitivity and specificity. However, false positives and false negatives can occur. Factors such as cross-reactivity with other antibodies and variations in immune response can impact accuracy. It's crucial to choose validated tests and interpret results in conjunction with clinical context.
For IgM detection, testing is most effective 7-14 days after symptom onset or suspected exposure. IgG antibodies typically take 2-3 weeks to develop, so testing for IgG is more reliable after this timeframe. It's best to consult with a healthcare professional to determine the optimal timing based on individual circumstances.
A positive IgG result indicates the presence of antibodies, suggesting some level of immunity. However, it doesn't guarantee complete protection from future infection. The level of antibodies, duration of protection, and potential for new variants all play a role. It is important to still practice preventative measures, like vaccination and hygiene, even with a positive result.
Test covid-19 igg igm provides valuable insights into past infection and immunity but shouldn't be the sole basis for public health decisions. It doesn't detect current infection, and antibody levels can vary. Combining serological data with PCR testing, vaccination rates, and epidemiological data provides a more comprehensive picture of disease transmission and risk.
Like any blood test, there is a small risk of discomfort, bruising, or infection at the puncture site. These risks are minimal. The test itself doesn’t pose any significant health risks. Individuals with specific medical conditions should consult their healthcare provider before undergoing any testing.
The understanding and application of test covid-19 igg igm have been instrumental in navigating the COVID-19 pandemic and continue to be vital for public health surveillance. The ability to detect past infection, assess immunity levels, and inform vaccination strategies has empowered individuals and communities to better protect themselves. The collaborative efforts of researchers, manufacturers, and healthcare professionals have resulted in increasingly accurate and accessible testing solutions.
Looking ahead, continued innovation in antibody detection technologies, coupled with data-driven approaches, will further enhance our ability to anticipate and respond to emerging infectious disease threats. By embracing these advancements and fostering global collaboration, we can build a more resilient and prepared world. For more information about our COVID-19 testing services, visit our website: www.prisesbio.com.